
www.invitrogen.com/cellculturebasics
B-087243 0110
CELL CULTURE BASICS
Handbook
Cell Culture Basics
Cell Culture Basics


Cell Culture Basics | i
Contents
Introduction. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1
Purpose of the Handbook. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .1
Introduction to Cell Culture . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2
What is Cell Culture? . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2
Finite vs Continuous Cell Line. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2
Culture Conditions . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2
Cryopreservation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2
Morphology of Cells in Culture . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3
Applications of Cell Culture. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3
Cell Culture Laboratory. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4
Safety . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4
Biosafety Levels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4
SDS. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5
Safety Equipment . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5
Personal Protective Equipment (PPE) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5
Safe Laboratory Practices. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5
Cell Culture Equipment . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6
Basic Equipment . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6
Expanded Equipment . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6
Additional Supplies . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6
Cell Culture Laboratory . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7
Aseptic Work Area. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7
Cell Culture Hood . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7
Cell Culture Hood Layout. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8
Incubator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9
Storage . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9
Cryogenic Storage . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10
Cell Counter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10

ii | Cell Culture Basics
Contents
Aseptic Technique. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11
Sterile Work Area. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11
Good Personal Hygiene . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11
Sterile Reagents and Media. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 12
Sterile Handling. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 12
Aseptic Technique Checklist. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 13
Biological Contamination . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14
Bacteria . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14
Yeasts . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 15
Molds . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 15
Viruses. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 16
Mycoplasma . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 16
Cross-Contamination. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 17
Using Antibiotics. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 17
Cell Culture Basics . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 18
Cell Lines . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 18
Selecting the Appropriate Cell Line . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 18
Acquiring Cell Lines . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 18
Culture Environment . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 19
Adherent vs Suspension Culture . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 19
Media . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 20
pH . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 21
CO
2
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 21
Temperature . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 21
Cell Morphology . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 22
Mammalian Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 22
Variations in Mammalian Cell Morphology . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 22
Morphology of 293 Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 23
Insect Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 24
Morphology of Sf21 Cells. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 24
Morphology of Sf9 Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 25

Cell Culture Basics | iii
Contents
Methods . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 26
Guidelines for Maintaining Cultured Cells. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 26
What is Subculture? . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 26
When to Subculture?. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 27
Media Recommendations for Common Cell Lines . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 28
Dissociating Adherent Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 30
TrypLE
™
Dissociation Enzymes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 30
Subculturing Adherent Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 31
Materials Needed . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 31
Protocol for Passaging Adherent Cells. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 31
Notes on Subculturing Adherent Insect Cells. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 32
Subculturing Suspension Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 33
Passaging Suspension Cultures . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 33
Suspension Culture Vessels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 33
Materials Needed . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 34
Protocol for Passaging Suspension Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 34
Notes on Subculturing Suspension Insect Cells. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 36
Freezing Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 37
Cryopreservation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 37
Guidelines for Cryopreservation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 37
Freezing Medium . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 38
Materials Needed . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 38
Protocol for Cryopreserving Cultured Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 39
Thawing Frozen Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 40
Guidelines for Thawing . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 40
Materials Needed . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 40
Protocol for Thawing Frozen Cells. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 40
Support Protocols . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 41
Counting Cells in a Hemacytometer . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 41
Trypan Blue Exclusion . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 42
Concentrating Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 42

iv | Cell Culture Basics
Appendix. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 43
Troubleshooting . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 43
Cell Culture Products . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 44
Cell Lines . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 44
Media for Mammalian Cell Culture . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 45
Media for Insect Cell Culture. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 46
Serum Products for Cell Culture. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 46
Laboratory Reagents for Cell Culture. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 47
Antibiotics and Antimycotics . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 48
Accessory Products for Cell Culture . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 49
Growth Factors and Purified Proteins . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 49
Transfection and Selection . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 50
Transfection Reagents. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 51
Additional Resources . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 52
Mammalian and Insect Cell Cultures . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 52
Cell and Tissue Analysis . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 52
Transfection Selection Tool . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 52
Safety Data Sheets . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 52
Certificate of Analysis . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 52
Technical Support . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 53

Introduction
Cell Culture Basics | 1
Purpose of the Handbook
Cell Culture Basics Companion Handbook is a supplement to the Cell Culture Basics
instructional videos available online at www.invitrogen.com/cellculturebasics.
The handbook and videos are intended as an introduction to cell culture basics, covering
topics such as getting familiar with the requirements of a laboratory dedicated to cell
culture experiments, laboratory safety, aseptic technique, and microbial contamination
of cell cultures, as well as providing basic methods for passaging, freezing, and thawing
cultured cells.
The information and guidelines presented in the handbook and the instructional videos
focus on cell lines (finite or continuous) and omit experiments and techniques concerning
primary cultures such as isolating and disaggregating tissues.
Note that while the basics of cell culture experiments share certain similarities, cell culture
conditions vary widely for each cell type. Deviating from the culture conditions required
for a particular cell type can result in different phenotypes being expressed; we therefore
recommend that you familiarize yourself with your cell line of interest, and closely follow
the instructions provided with each product you are using in your experiments.

2 | Cell Culture Basics
Part 1. Introduction
Introduction to Cell Culture
What is Cell
Culture?
Cell culture refers to the removal of cells from an animal or plant and their subsequent
growth in a favorable artificial environment. The cells may be removed from the tissue
directly and disaggregated by enzymatic or mechanical means before cultivation, or they
may be derived from a cell line or cell strain that has already been already established.
Primary Culture
Primary culture refers to the stage of the culture after the cells are isolated from the tissue
and proliferated under the appropriate conditions until they occupy all of the available
substrate (i.e., reach confluence). At this stage, the cells have to be subcultured (i.e.,
passaged) by transferring them to a new vessel with fresh growth medium to provide
more room for continued growth.
Cell Line
After the first subculture, the primary culture becomes known as a cell line or subclone.
Cell lines derived from primary cultures have a limited life span (i.e., they are finite; see
below), and as they are passaged, cells with the highest growth capacity predominate,
resulting in a degree of genotypic and phenotypic uniformity in the population.
Cell Strain
If a subpopulation of a cell line is positively selected from the culture by cloning or some
other method, this cell line becomes a cell strain. A cell strain often acquires additional
genetic changes subsequent to the initiation of the parent line.
Finite vs
Continuous
Cell Line
Normal cells usually divide only a limited number of times before losing their ability to
proliferate, which is a genetically determined event known as senescence; these cell lines
are known as finite. However, some cell lines become immortal through a process called
transformation, which can occur spontaneously or can be chemically or virally induced.
When a finite cell line undergoes transformation and acquires the ability to divide
indefinitely, it becomes a continuous cell line.
Culture
Conditions
Culture conditions vary widely for each cell type, but the artificial environment in which
the cells are cultured invariably consists of a suitable vessel containing a substrate or
medium that supplies the essential nutrients (amino acids, carbohydrates, vitamins,
minerals), growth factors, hormones, and gases (O
2
, CO
2
), and regulates the physico-
chemical environment (pH, osmotic pressure, temperature). Most cells are anchorage-
dependent and must be cultured while attached to a solid or semi-solid substrate
(adherent or monolayer culture), while others can be grown floating in the culture
medium (suspension culture).
Cryopreservation If a surplus of cells are available from subculturing, they should be treated with the
appropriate protective agent (e.g., DMSO or glycerol) and stored at temperatures below
–130°C (cryopreservation) until they are needed. For more information on subculturing
and cryopreserving cells, refer to the Guidelines for Maintaining Cultured Cells,
page page 26–page page 39.

Cell Culture Basics | 3
Part 1. Introduction
Morphology of
Cells in Culture
Cells in culture can be divided in to three basic categories based on their shape and
appearance (i.e., morphology).
• Fibroblastic (or fibroblast-like) cells are bipolar or multipolar, have elongated shapes,
and grow attached to a substrate.
• Epithelial-like cells are polygonal in shape with more regular dimensions, and grow
attached to a substrate in discrete patches.
• Lymphoblast-like cells are spherical in shape and usually grown in suspension
without attaching to a surface.
Applications of
Cell Culture
Cell culture is one of the major tools used in cellular and molecular biology, providing
excellent model systems for studying the normal physiology and biochemistry of cells
(e.g., metabolic studies, aging), the effects of drugs and toxic compounds on the cells,
and mutagenesis and carcinogenesis. It is also used in drug screening and development,
and large scale manufacturing of biological compounds (e.g., vaccines, therapeutic
proteins). The major advantage of using cell culture for any of the these applications is
the consistency and reproducibility of results that can be obtained from using a batch of
clonal cells.

4 | Cell Culture Basics
Cell Culture Laboratory
Safety
In addition to the safety risks common to most everyday work places such as electrical
and fire hazards, a cell culture laboratory has a number of specific hazards associated with
handling and manipulating human or animal cells and tissues, as well as toxic, corrosive,
or mutagenic solvents and reagents. The most common of these hazards are accidental
punctures with syringe needles or other contaminated sharps, spills and splashes
onto skin and mucous membranes, ingestion through mouth pipetting, and inhalation
exposures to infectious aerosols.
The fundamental objective of any biosafety program is to reduce or eliminate exposure of
laboratory workers and the outside environment to potentially harmful biological agents.
The most important element of safety in a cell culture laboratory is the strict adherence
to standard microbiological practices and techniques.
Biosafety Levels The regulations and recommendations for biosafety in the United States are contained
in the document Biosafety in Microbiological and Biomedical Laboratories, prepared by the
Centers for Disease Control (CDC) and the National Institutes of Health (NIH), and
published by the U.S. Department of Health and Human Services. The document defines
four ascending levels of containment, referred to as biosafety levels 1 through 4, and
describes the microbiological practices, safety equipment, and facility safeguards for the
corresponding level of risk associated with handling a particular agent.
Biosafety Level 1 (BSL-1)
BSL-1 is the basic level of protection common to most research and clinical laboratories,
and is appropriate for agents that are not known to cause disease in normal, healthy
humans.
Biosafety Level 2 (BSL-2)
BSL-2 is appropriate for moderate-risk agents known to cause human disease of varying
severity by ingestion or through percutaneous or mucous membrane exposure. Most cell
culture labs should be at least BSL-2, but the exact requirements depend upon the cell line
used and the type of work conducted.
Biosafety Level 3 (BSL-3)
BSL-3 is appropriate for indigenous or exotic agents with a known potential for aerosol
transmission, and for agents that may cause serious and potentially lethal infections.
Biosafety Level 4 (BSL-4)
BSL-4 is appropriate for exotic agents that pose a high individual risk of life-threatening
disease by infectious aerosols and for which no treatment is available. These agents are
restricted to high containment laboratories.
For more information about the biosafety level guidelines, refer to Biosafety in
Microbiological and Biomedical Laboratories, 5
th
Edition, which is available for downloading at
www.cdc.gov/od/ohs/biosfty/bmbl5/bmbl5toc.htm.

Cell Culture Basics | 5
Part 2. Cell Culture Laboratory
Safety Data Sheet
(SDS)
Safety Data Sheet (SDS), also referred to as Material Safety Data Sheet (MSDS), is a form
containing information regarding the properties of a particular substance. The SDS
includes physical data such as melting point, boiling point, and flash point, information
on the substance’s toxicity, reactivity, health effects, storage, and disposal, as well as
recommended protective equipment and procedures for handling spills.
The SDSs for all Invitrogen products are available at www.invitrogen.com/sds.
Safety Equipment Safety equipment in a cell culture laboratory includes primary barriers such as biosafety
cabinets, enclosed containers, and other engineering controls designed to remove or
minimize exposure to hazardous materials, as well as personal protective equipment
(PPE) that is often used in conjunction with the primary barriers. The biosafety cabinet
(i.e., cell culture hood) is the most important equipment to provide containment of
infectious splashes or aerosols generated by many microbiological procedures as well as
to prevent contamination of your own cell culture. For more information, see Cell Culture
Hood, page 7.
Personal Protective
Equipment (PPE)
Personal protective equipment (PPE) form an immediate barrier between the personnel
and the hazardous agent, and they include items for personal protection such as gloves,
laboratory coats and gowns, shoe covers, boots, respirators, face shields, safety glasses,
or goggles. They are often used in combination with biosafety cabinets and other devices
that contain the agents or materials being handled. We recommend that you consult your
institution’s guidelines for the appropriate use of PPE in your laboratory.
Safe Laboratory
Practices
The following recommendations are simply guidelines for safe laboratory practices, and
they should not be interpreted as a complete code of practice. Consult your institution’s
safety committee and follow local rules and regulations pertaining to laboratory safety.
For more information on standard microbiological practices and for specific biosafety
level guidelines, refer to Biosafety in Microbiological and Biomedical Laboratories, 5
th
Edition at
www.cdc.gov/od/ohs/biosfty/bmbl5/bmbl5toc.htm.
• Always wear appropriate personal protective equipment. Change gloves when
contaminated, and dispose of used gloves with other contaminated laboratory waste.
• Wash your hands after working with potentially hazardous materials and before
leaving the laboratory.
• Do not eat, drink, smoke, handle contact lenses, apply cosmetics, or store food for
human consumption in the laboratory.
• Follow the institutional policies regarding safe handling of sharps (i.e., needles,
scalpels, pipettes, and broken glassware).
• Take care to minimize the creation of aerosols and/or splashes.
• Decontaminate all work surfaces before and after your experiments, and immediately
after any spill or splash of potentially infectious material with an appropriate
disinfectant. Clean laboratory equipment routinely, even if it is not contaminated.
• Decontaminate all potentially infectious materials before disposal.
• Report any incidents that may result in exposure to infectious materials to appropriate
personnel (e.g., laboratory supervisor, safety officer).

6 | Cell Culture Basics
Part 2. Cell Culture Laboratory
Cell Culture Equipment
The specific requirements of a cell culture laboratory depend mainly on the type of
research conducted; for example, the needs of mammalian cell culture laboratory
specializing in cancer research is quite different from that of an insect cell culture
laboratory that focuses on protein expression. However, all cell culture laboratories have
the common requirement of being free from pathogenic microorganisms (i.e., asepsis), and
share some of the same basic equipment that is essential for culturing cells.
This section lists the equipment and supplies common to most cell culture laboratories,
as well as beneficial equipment that allows the work to be performed more efficiently
or accurately, or permits wider range of assays and analyses. Note that this list is not
all inclusive; the requirements for any cell culture laboratory depend the type of work
conducted.
Basic Equipment • Cell culture hood (i.e., laminar-flow hood or biosafety cabinet)
• Incubator (humid CO
2
incubator recommended)
• Water bath
• Centrifuge
• Refrigerator and freezer (–20°C)
• Cell counter (e.g., Countess
®
Automated Cell Counter or hemacytometer)
• Inverted microscope
• Liquid nitrogen (N
2
) freezer or cryostorage container
• Sterilizer (i.e., autoclave)
Expanded Equipment • Aspiration pump (peristaltic or vacuum)
• pH meter
• Confocal microscope
• Flow cytometer
Additional Supplies • Cell culture vessels (e.g., flasks, Petri dishes, roller bottles, multi-well plates)
• Pipettes and pipettors
• Syringes and needles
• Waste containers
• Media, sera, and reagents
• Cells

Cell Culture Basics | 7
Part 2. Cell Culture Laboratory
Cell Culture Laboratory
Aseptic Work Area The major requirement of a cell culture laboratory is the need to maintain an aseptic
work area that is restricted to cell culture work. Although a separate tissue culture room
is preferred, a designated cell culture area within a larger laboratory can still be used
fort sterile handling, incubation, and storage of cell cultures, reagents, and media. The
simplest and most economical way to provide aseptic conditions is to use a cell culture
hood (i.e., biosafety cabinet).
Cell Culture Hood The cell culture hood provides an aseptic work area while allowing the containment of
infectious splashes or aerosols generated by many microbiological procedures. Three
kinds of cell culture hoods, designated as Class I, II and III, have been developed to meet
varying research and clinical needs.
Classes of Cell Culture Hoods
ClassI cell culture hoods offer significant levels of protection to laboratory personnel and
to the environment when used with good microbiological techniques, but they do not
provide cultures protection from contamination. They are similar in design and air flow
characteristics to chemical fume hoods.
ClassII cell culture hoods are designed for work involving BSL-1, 2, and 3 materials, and
they also provide an aseptic environment necessary for cell culture experiments. A Class
II biosafety cabinet should be used for handling potentially hazardous materials (e.g.,
primate-derived cultures, virally infected cultures, radioisotopes, carcinogenic or toxic
reagents).
Class III biosafety cabinets are gas-tight, and they provide the highest attainable level of
protection to personnel and the environment. A Class III biosafety cabinet is required for
work involving known human pathogens and other BSL-4 materials.
Air-Flow Characteristics of Cell Culture Hoods
Cell culture hoods protect the working environment from dust and other airborn
contaminants by maintaining a constant, unidirectional flow of HEPA-filtered air over the
work area. The flow can be horizontal, blowing parallel to the work surface, or it can be
vertical, blowing from the top of the cabinet onto the work surface.
Depending on its design, a horizontal flow hood provides protection to the culture (if the
air flowing towards the user) or to the user (if the air is drawn in through the front of the
cabinet by negative air pressure inside). Vertical flow hoods, on the other hand, provide
significant protection to the user and the cell culture.
Clean Benches
Horizontal laminar flow or vertical laminar flow “clean benches” are not biosafety
cabinets; these pieces of equipment discharge HEPA-filtered air from the back of the
cabinet across the work surface toward the user, and they may expose the user to
potentially hazardous materials. These devices only provide product protection. Clean
benches can be used for certain clean activities, such as the dust-free assembly of sterile
equipment or electronic devices, and they should never be used when handling cell
culture materials or drug formulations, or when manipulating potentially infectious
materials.
For more information on the selection, installation, and use of biosafety cabinets, refer
to Biosafety in Microbiological and Biomedical Laboratories, 5
th
Edition, which is available for
downloading at www.cdc.gov/od/ohs/biosfty/bmbl5/bmbl5toc.htm.

8 | Cell Culture Basics
Part 2. Cell Culture Laboratory
Cell Culture Hood
Layout
A cell culture hood should be large enough to be used by one person at a time, be easily
cleanable inside and outside, have adequate lighting, and be comfortable to use without
requiring awkward positions. Keep the work space in the cell culture hood clean and
uncluttered, and keep everything in direct line of sight. Disinfect each item placed in the
cell culture hood by spraying them with 70% ethanol and wiping clean.
The arrangement of items within the cell culture hood usually adheres to the following
right-handed convention, which can be modified to include additional items used in
specific applications.
• A wide, clear work space in the center with your cell culture vessels
• Pipettor in the front right, where it can be reached easily
• Reagents and media in the rear right to allow easy pipetting
• Tube rack in the rear middle holding additional reagents
• Small container in the rear left to hold liquid waste
Waste Liquid
Reagents and Media
Cell Culture Flasks
Tube Rack
70%
Ethanol
spray
Waste Container
Pipettor
Wrapped
Disposable
Pipettes
DMEM
DPBS
Figure 2.1. The basic layout of a cell culture hood for right-handed workers. Left-handed workers may switch the positions of the items laid
out on the work surface.

Cell Culture Basics | 9
Part 2. Cell Culture Laboratory
Incubator The purpose of the incubator is to provide the appropriate environment for cell
growth. The incubator should be large enough for your laboratory needs, have forced-
air circulation, and should have temperature control to within ±0.2°C. Stainless steel
incubators allow easy cleaning and provide corrosion protection, especially if humid
air is required for incubation. Although the requirement for aseptic conditions in a cell
culture incubator is not as stringent as that in a cell culture hood, frequent cleaning of the
incubator is essential to avoid contamination of cell cultures.
Types of Incubators
There are two basic types of incubators, dry incubators and humid CO
2
incubators. Dry
incubators are more economical, but require the cell cultures to be incubated in sealed
flasks to prevent evaporation. Placing a water dish in a dry incubator can provide some
humidity, but they do not allow precise control of atmospheric conditions in the incubator.
Humid CO
2
incubators are more expensive, but allow superior control of culture
conditions. They can be used to incubate cells cultured in Petri dishes or multi-well plates,
which require a controlled atmosphere of high humidity and increased CO
2
tension.
Storage A cell culture laboratory should have storage areas for liquids such as media and reagents,
for chemicals such as drugs and antibiotics, for consumables such as disposable pipettes,
culture vessels, and gloves, for glassware such as media bottles and glass pipettes, for
specialized equipment, and for tissues and cells.
Glassware, plastics, and specialized equipment can be stored at ambient temperature
on shelves and in drawers; however, it is important to store all media, reagents, and
chemicals according to the instructions on the label.
Some media, reagents, and chemicals are sensitive to light; while their normal laboratory
use under lighted conditions is tolerated, they should be stored in the dark or wrapped in
aluminum foil when not in use.
Refrigerators
For small cell culture laboratories, a domestic refrigerator (preferably one without a
autodefrost freezer) is an adequate and inexpensive piece of equipment for storing
reagents and media at 2–8°C. For larger laboratories, a cold room restricted to cell culture
is more appropriate. Make sure that the refrigerator or the cold room is cleaned regularly
to avoid contamination.
Freezers
Most cell culture reagents can be stored at –5°C to –20°C; therefore an ultradeep freezer
(i.e., a –80°C freezer) is optional for storing most reagents. A domestic freezer is a cheaper
alternative to a laboratory freezer. While most reagents can withstand temperature
oscillations in an autodefrost (i.e., self-thawing) freezer, some reagents such as antibiotics
and enzymes should be stored in a freezer that does not autodefrost.

10 | Cell Culture Basics
Part 2. Cell Culture Laboratory
Cryogenic Storage Cell lines in continuous culture are likely to suffer from genetic instability as their passage
number increases; therefore, it is essential to prepare working stocks of the cells and
preserve them in cryogenic storage (for more information, see Freezing Cells,
page 37). Do not store cells in –20°C or –80°C freezers, because their viability quickly
decreases when they are stored at these temperatures.
There are two main types of liquid-nitrogen storage systems, vapor phase and liquid
phase, which come as wide-necked or narrow-necked storage containers. Vapor phase
systems minimize the risk of explosion with cryostorage tubes, and are required for
storing biohazardous materials, while the liquid phase systems usually have longer static
holding times, and are therefore more economical.
Narrow-necked containers have a slower nitrogen evaporation rate and are more
economical, but wide-necked containers allow easier access and have a larger storage
capacity.
Cell Counter A cell counter is essential for quantitative growth kinetics, and a great advantage when
more than two or three cell lines are cultured in the laboratory.
The Countess
®
Automated Cell Counter is a bench-top instrument designed to measure
cell count and viability (live, dead, and total cells) accurately and precisely in less than
a minute per sample, using the standard Trypan Blue uptake technique. Using the
same amount of sample that you currently use with the hemacytometer, the Countess
®
Automated Cell Counter takes less than a minute per sample for a typical cell count and is
compatible with a wide variety of eukaryotic cells.

Cell Culture Basics | 11
Part 2. Cell Culture Laboratory
Aseptic Technique
Introduction Successful cell culture depends heavily on keeping the cells free from contamination by
microorganisms such as bacterial, fungi, and viruses. Non-sterile supplies, media, and
reagents, airborne particles laden with microorganisms, unclean incubators, and dirty
work surfaces are all sources of biological contamination.
Aseptic technique, designed to provide a barrier between the microrganisms in the
environment and the sterile cell culture, depends upon a set of procedures to reduce the
probability of contamination from these sources. The elements of aseptic technique are a
sterile work area, good personal hygiene, sterile reagents and media, and sterile handling.
Sterile Work Area The simplest and most economical way to reduce contamination from airborne particles
and aerosols (e.g., dust, spores, shed skin, sneezing) is to use a cell culture hood.
• The cell culture hood should be properly set up and be located in an area that is
restricted to cell culture that is free from drafts from doors, windows, and other
equipment, and with no through traffic.
• The work surface should be uncluttered and contain only items required for a
particular procedure; it should not be used as a storage area.
• Before and after use, the work surface should be disinfected thoroughly, and the
surrounding areas and equipment should be cleaned routinely.
• For routine cleaning, wipe the work surface with 70% ethanol before and during work,
especially after any spillage.
• You may use ultraviolet light to sterilize the air and exposed work surfaces in the cell
culture hood between uses.
• Using a Bunsen burner for flaming is not necessary nor recommended in a cell culture
hood.
• Leave the cell culture hood running at all times, turning them off only when they will
not be used for extended periods of time.
Good Personal
Hygiene
Wash your hands before and after working with cell cultures. In addition to protecting
you from hazardous materials, wearing personal protective equipment also reduces the
probability of contamination from shed skin as well as dirt and dust from your clothes.
Sterile Reagents and
Media
Commercial reagents and media undergo strict quality control to ensure their sterility, but
they can become contaminated while handling. Follow the guidelines below for sterile
handling to avoid contaminating them. Always sterilize any reagents, media, or solutions
prepared in the laboratory using the appropriate sterilization procedure (e.g., autoclave,
sterile filter).

12 | Cell Culture Basics
Part 2. Cell Culture Laboratory
Sterile Handling • Always wipe your hands and your work area with 70% ethanol.
• Wipe the outside of the containers, flasks, plates, and dishes with 70% ethanol before
placing them in the cell culture hood.
• Avoid pouring media and reagents directly from bottles or flasks.
• Use sterile glass or disposable plastic pipettes and a pipettor to work with liquids, and
use each pipette only once to avoid cross contamination. Do not unwrap sterile pipettes
until they are to be used. Keep your pipettes at your work area.
• Always cap the bottles and flasks after use and seal multi-well plates with tape or place
them in resealable bags to prevent microorganisms and airborn contaminants from
gaining entry.
• Never uncover a sterile flask, bottle, petri dish, etc. until the instant you are ready to
use it and never leave it open to the environment. Return the cover as soon as you are
finished.
• If you remove a cap or cover, and have to put it down on the work surface, place the
cap with opening facing down.
• Use only sterile glassware and other equipment.
• Be careful not to talk, sing, or whistle when you are performing sterile procedures.
• Perform your experiments as rapidly as possible to minimize contamination.

Cell Culture Basics | 13
Part 2. Cell Culture Laboratory
Work Area
Is the cell culture hood properly set up?
Is the cell culture hood in an area free from drafts and through trac?
Is the work surface uncluttered, and does it contain only items required for your
experiment?
Did you wipe the work surface with 70% ethanol before work?
Are you routinely cleaning and sterilizing your incubators, refrigerators, freezers, and other
laboratory equipment?
Personal Hygiene
Did you wash your hands?
Are you wearing personal protective equipment?
If you have long hair, is it tied in the back?
Are you using a pipettor to work with liquids?
Reagents and Media
Have you sterilized any reagents, media, and solutions you have prepared in the laboratory
using the appropriate procedure?
Did you wipe the outside of the bottles, asks, and plates with 70% ethanol before placing
them on your work surface?
Are all your bottles, asks, and other containers capped when not in use?
Are all your plates stored in sterile resealable bags?
Does any of your reagents look cloudy? Contaminated? Do they contain oating particles?
Have foul smell? Unusual color? If yes, did you decontaminated and discarded them?
Handling
Are you working slowly and deliberately, mindful of aseptic technique?
Did you wipe the surfaces of all the items including pipettor, bottles, and asks with 70%
ethanol before placing them in the cell culture hood?
Are placing the caps or covers face down on the work area?
Are you using sterile glass pipettes or sterile disposable plastic pipettes to manipulate all
liquids?
Are you using a sterile pipette only once to avoid cross contamination?
Are you careful not to touch the pipette tip to anything non-sterile, including the outside
edge of the bottle threads?
Did you mop up any spillage immediately, and wiped the area with 70% ethanol?
Aseptic Technique Checklist
The following checklist provides a concise list of suggestions and procedures to guide you
to achieve a solid aseptic technique. For an in-depth review of aseptic technique, refer to
Culture of Animal Cells: A Manual of Basic Technique (Freshney, 2000).

14 | Cell Culture Basics
Part 2. Cell Culture Laboratory
Biological Contamination
Introduction Contamination of cell cultures is easily the most common problem encountered in
cell culture laboratories, sometimes with very serious consequences. Cell culture
contaminants can be divided into two main categories, chemical contaminants such
as impurities in media, sera, and water, endotoxins, plasticizers, and detergents, and
biological contaminants such as bacteria, molds, yeasts, viruses, mycoplasma, as well as
cross contamination by other cell lines. While it is impossible to eliminate contamination
entirely, it is possible to reduce its frequency and seriousness by gaining a thorough
understanding of their sources and by following good aseptic technique. This section
provides an overview of major types of biological contamination.
Bacteria Bacteria are a large and ubiquitous group of unicellular microorganisms. They are
typically a few micrometers in diameters, and can have a variety of shapes, ranging
from spheres to rods and spirals. Because of their ubiquity, size, and fast growth rates,
bacteria, along with yeasts and molds, are the most commonly encountered biological
contaminants in cell culture. Bacterial contamination is easily detected by visual
inspection of the culture within a few days of it becoming infected; infected cultures
usually appear cloudy (i.e., turbid), sometimes with a thin film on the surface. Sudden
drops in the pH of the culture medium is also frequently encountered. Under a low-
power microscope, the bacteria appear as tiny, moving granules between the cells, and
observation under a high-power microscope can resolve the shapes of individual bacteria.
The simulated images below show an adherent 293 cell culture contaminated with E. coli.
Figure 2.2. Simulated phase contrast images of adherent 293 cells contaminated with E. coli. The spaces between
the adherent cells show tiny, shimmering granules under low power microscopy, but the individual bacteria are not
easily distinguishable (panel A). Further magnification of the area enclosed by the black square resolves the individual
E. coli cells, which are typically rod-shaped and are about 2 μm long and 0.5 μm in diameter. Each side of the black
square in panel A is 100 μm.
A B

Cell Culture Basics | 15
Part 2. Cell Culture Laboratory
Yeasts Yeasts are unicellular eukaryotic microorganisms in the kingdom of Fungi, ranging in
size from a few micrometers (typically) up to 40 micrometers (rarely). Like bacterial
contamination, cultures contaminated with yeasts become turbid, especially if the
contamination is in an advanced stage. There is very little change in the pH of the culture
contaminated by yeasts until the contamination becomes heavy, at which stage the pH
usually increases. Under microscopy, yeast appear as individual ovoid or spherical
particles, that may bud off smaller particles. The simulated image below shows adherent
293 cell culture 24 hours after plating that is infected with yeast.
Molds Molds are eukaryotic microorganisms in the kingdom of Fungi that grow as multicellular
filaments called hyphae. A connected network of these multicellular filaments contain
genetically identical nuclei, and are referred to as a colony or mycelium. Similar to yeast
contamination, the pH of the culture remains stable in the initial stages of contamination,
then rapidly increases as the culture become more heavily infected and becomes turbid.
Under microscopy, the mycelia usually appear as thin, wisp-like filaments, and sometimes
as denser clumps of spores. Spores of many mold species can survive extremely harsh and
inhospitable environments in their dormant stage, only to become activated when they
encounter suitable growth conditions.
Figure 2.3. Simulated phase contrast images of 293 cells in adherent culture that is contaminated with yeast.
The contaminating yeast cells appear as ovoid particles, budding off smaller particles as they replicate.

16 | Cell Culture Basics
Part 2. Cell Culture Laboratory
Viruses Viruses are microscopic infectious agents that take over the host cells machinery to
reproduce. Their extremely small size makes them very difficult to detect in culture, and
to remove them from reagents used in cell culture laboratories. Because most viruses
have very stringent requirements for their host, they usually do not adversely effect cell
cultures from species other than their host. However, using virally infected cell cultures
can present a serious health hazard to the laboratory personnel, especially if human or
primate cells are cultured in the laboratory. Viral infection of cell cultures can be detected
by electron microscopy, immunostaining with a panel of antibodies, ELISA assays, or PCR
with appropriate viral primers.
Mycoplasma Mycoplasma are simple bacteria that lack a cell wall, and they are considered the smallest
self-replicating organism. Because of their extremely small size (typically less than one
micrometer), mycoplasma are very difficult to detect until they achieve extremely high
densities and cause the cell culture to deteriorate; until then, there are often no visible
signs of infection. Some slow growing mycoplasma may persists in culture without
causing cell death, but they can alter the behavior and metabolism of the host cells in the
culture. Chronic mycoplasma infections might manifest themselves with decreased rate
of cell proliferation, reduced saturation density, and agglutination in suspension cultures;
however, the only assured way of detecting mycoplasma contamination is by testing
the cultures periodically using fluorescent staining (e.g., Hoechst33258), ELISA, PCR,
immunostaining, autoradiography, or microbiological assays.
Figure 2.4. Photomicrographs of mycoplasma-free cultured cells (panel A) and cells infected with mycoplasma (panels B and C). The cultures were tested
using the MycoFluor™ Mycoplasma Detection Kit, following the kit protocols. In fixed cells, the MycoFluor™ reagent has access to the cell nuclei, which are
intensely stained with the reagent, but the absence of fluorescent extranuclear objects indicates that the culture is free from mycoplasma contamination
(panel A). In fixed cells infected with mycoplasma, the MycoFluor™ reagent stains both the nuclei and the mycoplasma, but the intense relative fluorescence
of the nuclei obscure the mycoplasma on or near the nuclei. However, the mycoplasma separated from the bright nuclei are readily visible (panel B).
In live cells, the MycoFluor™ reagent does not have access to the nuclei, but readily stains the mycoplasma associated with the outside of cells (panel
C). The images were obtained using 365 nm excitation and a 100/1.3 Plan Neoflaur® (Zeiss) objective lens coupled with a 450 ± 30 nm bandpass filter.
A B C

Cell Culture Basics | 17
Part 2. Cell Culture Laboratory
Cross-Contamination While not as common as microbial contamination, extensive cross-contamination of many
cell lines with HeLa and other fast growing cell lines is a clearly-established problem with
serious consequences. Obtaining cell lines from reputable cell banks, periodically checking
the characteristics of the cell lines, and practicing good aseptic technique are practices that
will help you avoid cross-contamination. DNA fingerprinting, karyotype analysis, and
isotype analysis can confirm the presence or absence of cross-contamination in your cell
cultures.
Using Antibiotics Antibiotics should not be used routinely in cell culture, because their continuous
use encourages the development of antibiotic resistant strains and allows low-level
contamination to persist, which can develop into full-scale contamination once the
antibiotic is removed from media, and may hide mycoplasma infections and other cryptic
contaminants. Further, some antibiotics might cross react with the cells and interfere with
the cellular processes under investigation.
Antibiotics should only be used as a last resort and only for short term applications,
and they should be removed from the culture as soon as possible. If they are used in the
long term, antibiotic-free cultures should be maintained in parallel as a control for cryptic
infections.

18 | Cell Culture Basics
Cell Culture Basics
This section provides information on the fundamentals of cell culture, including the
selection of the appropriate cell line for your experiments, media requirements for cell
culture, adherent versus suspension culture, and morphologies of continuous cell lines
available from Invitrogen.
Note that the following information is an introduction to the basics of cell culture, and it
is intended as a starting point in your investigations. For more in-depth information, we
recommend that you consult published literature and books, as well as the manuals and
product information sheets provided with the products you are using.
Cell Lines
Selecting the
Appropriate Cell
Line
Consider the following criteria for selecting the appropriate cell line for your experiments:
• Species: Non-human and non-primate cell lines usually have fewer biosafety
restrictions, but ultimately your experiments will dictate whether to use species-
specific cultures or not.
• Functional characteristics: What is the purpose of your experiments? For example,
liver- and kidney-derived cell lines may be more suitable for toxicity testing.
• Finite or continuous: While choosing from finite cell lines may give you more options
to express the correct functions, continuous cell lines are often easier to clone and
maintain.
• Normal or transformed: Transformed cell lines usually have an increased growth
rate and higher plating efficiency, are continuous, and require less serum in media,
but they have undergone a permanent change in their phenotype through a genetic
transformation.
• Growth conditions and characteristics: What are your requirements with respect
to growth rate, saturation density, cloning efficiency, and the ability to grow in
suspension? For example, to express a recombinant protein in high yields, you might
want to choose a cell line with a fast growth rate and an ability to grow in suspension.
• Other criteria: If you are using a finite cell line, are there sufficient stocks available?
Is the cell line well-characterized, or do you have to perform the validation yourself?
If you are using an abnormal cell line, do you have an equivalent normal cell line that
you can use as a control? Is the cell line stable? If not, how easy it is to clone it and
generate sufficient frozen stocks for your experiments?
Acquiring Cell Lines You may establish your own culture from primary cells, or you may choose to buy
established cell cultures from commercial or non-profit suppliers (i.e., cell banks).
Reputable suppliers provide high quality cell lines that are carefully tested for
their integrity and to ensure that the culture is free from contaminants. We advise
against borrowing cultures from other laboratories because they carry a high risk of
contamination. Regardless of their source, make sure that all new cell lines are tested for
mycoplasma contamination before you begin to use them.
Invitrogen offers a variety of primary cultures and established cell lines, reagents, media,
sera, and growth factors for your cell culture experiments. The Appendix section contains
a list of the more commonly used cell lines available from Invitrogen (see page page 44). For
more information on Invitrogen and GIBCO
®
products, refer to www.invitrogen.com.

Cell Culture Basics | 19
Part 3. Basic Cell Culture
Culture Environment
One of the major advantages of cell culture is the ability to manipulate the physico-
chemical (i.e., temperature, pH, osmotic pressure, O
2
and CO
2
tension) and the
physiological environment (i.e., hormone and nutrient concentrations) in which the cells
propagate. With the exception of temperature, the culture environment is controlled by
the growth media.
While the physiological environment of the culture is not as well defined as its physico-
chemical environment, a better understanding of the components of serum, the
identification of the growth factors necessary for proliferation, and a better appreciation
of the microenvironment of cells in culture (i.e., cell-cell interactions, diffusion of gases,
interactions with the matrix) now allow the culture of certain cell lines in serum-free
media.
Adherent vs
Suspension Culture
There are two basic systems for growing cells in culture, as monolayers on an artificial
substrate (i.e., adherent culture) or free-floating in the culture medium (suspension
culture). The majority of the cells derived from vertebrates, with the exception of
hematopoietic cell lines and a few others, are anchorage-dependent and have to be
cultured on a suitable substrate that is specifically treated to allow cell adhesion and
spreading (i.e., tissue-culture treated). However, many cell lines can also be adapted for
suspension culture. Similarly, most of the commercially available insect cell lines grow
well in monolayer or suspension culture. Cells that are cultured in suspension can be
maintained in culture flasks that are not tissue-culture treated, but as the culture volume
to surface area is increased beyond which adequate gas exchange is hindered (usually
0.2–0.5 mL/cm
2
), the medium requires agitation. This agitation is usually achieved with a
magnetic stirrer or rotating spinner flasks.
Adherent Culture Suspension Culture
Appropriate for most cell types, including
primary cultures.
Appropriate for cells adapted to suspension
culture and a few other cell lines that are
nonadhesive (e.g., hematopoietic).
Requires periodic passaging, but allows easy
visual inspection under inverted microscope.
Easier to passage, but requires daily cell counts
and viability determination to follow growth
patterns; culture can be diluted to stimulate
growth.
Cells are dissociated enzymatically (e.g.,
TrypLE™ Express, trypsin) or mechanically.
Does not require enzymatic or mechanical
dissociation.
Growth is limited by surface area, which may
limit product yields.
Growth is limited by concentration of cells in
the medium, which allows easy scale-up.
Requires tissue-culture treated vessel.
Can be maintained in culture vessels that
are not tissue-culture treated, but requires
agitation (i.e., shaking or stirring) for adequate
gas exchange.
Used for cytology, harvesting products
continuously, and many research applications.
Used for bulk protein production, batch
harvesting, and many research applications.

20 | Cell Culture Basics
Part 3. Basic Cell Culture
Media The culture medium is the most important component of the culture environment,
because it provides the necessary nutrients, growth factors, and hormones for cell growth,
as well as regulating the pH and the osmotic pressure of the culture.
Although initial cell culture experiments were performed using natural media obtained
from tissue extracts and body fluids, the need for standardization, media quality, and
increased demand led to the development of defined media. The three basic classes of
media are basal media, reduced-serum media, and serum-free media, which differ in
their requirement for supplementation with serum.
Serum is vitally important as a source of growth and adhesion factors, hormones, lipids
and minerals for the culture of cells in basal media. In addition, serum also regulates
cell membrane permeability and serves as a carrier for lipids, enzymes, micronutrients,
and trace elements into the cell. However, using serum in media has a number of
disadvantages including high cost, problems with standardization, specificity, variability,
and unwanted effects such as stimulation or inhibition of growth and/or cellular function
on certain cell cultures. If the serum is not obtained from reputable source, contamination
can also pose a serious threat to successful cell culture experiments. To address this threat,
all Invitrogen and GIBCO
®
products, including sera, are tested for contamination and
guaranteed for their quality, safety, consistency, and regulatory compliance.
Basal Media
The majority of cell lines grow well in basal media, which contain amino acids, vitamins,
inorganic salts, and a carbon source such as glucose, but these basal media formulations
must be further supplemented with serum.
Reduced-Serum Media
Another strategy to reduce the undesired effects of serum in cell culture experiments is to
use reduced-serum media. Reduced-serum media are basal media formulations enriched
with nutrients and animal-derived factors, which reduce the amount of serum that is
needed.
Serum-Free Media
Serum-free media (SFM) circumvents issues with using animal sera by replacing the
serum with appropriate nutritional and hormonal formulations. Serum-free media
formulations exist for many primary cultures and cell lines, including recombinant
protein producing lines of Chinese Hamster Ovary (CHO), various hybridoma cell lines,
the insect lines Sf9 and Sf21 (Spodoptera frugiperda), and for cell lines that act as hosts
for viral production (e.g., 293, VERO, MDCK, MDBK), and others. One of the major
advantages of using serum-free media is the ability to make the medium selective for
specific cell types by choosing the appropriate combination of growth factors. The table
below lists the advantages and disadvantages of serum-free media.
Advantages Disadvantages
• Increased denition
• More consistent performance
• Easier purication and downstream
processing
• Precise evaluation of cellular functions
• Increased productivity
• Better control over physiological response
• Enhanced detection of cellular mediators
• Requirement for cell type-specic media
formulations
• Need for higher degree of reagent purity
• Slower growth

Cell Culture Basics | 21
Part 3. Basic Cell Culture
Invitrogen offers a wide range of classical basal media, reduced-serum media, and serum-
free media, as well as sera, growth factors, supplements, antibiotics, and reagents for your
cell culture experiments. The Appendix section contains a list of the more commonly used
cell culture products available from Invitrogen. For more information on Invitrogen and
GIBCO
®
cell culture products, refer to www.invitrogen.com.
pH Most normal mammalian cell lines grow well at pH 7.4, and there is very little variability
among different cell strains. However, some transformed cell lines have been shown to
grow better at slightly more acidic environments (pH 7.0–7.4), and some normal fibroblast
cell lines prefer slightly more basic environments (pH 7.4–7.7). Insect cell lines such as Sf9
and Sf21 grow optimally at pH 6.2.
CO
2
The growth medium controls the pH of the culture and buffers the cells in culture against
changes in the pH. Usually, this buffering is achieved by including an organic (e.g.,
HEPES) or CO
2
-bicarbonate based buffer. Because the pH of the medium is dependent
on the delicate balance of dissolved carbon dioxide (CO
2
) and bicarbonate (HCO
3
–
),
changes in the atmospheric CO
2
can alter the pH of the medium. Therefore, it is necessary
to use exogenous CO
2
when using media buffered with a CO
2
-bicarbonate based buffer,
especially if the cells are cultured in open dishes or transformed cell lines are cultured at
high concentrations. While most researchers usually use 5–7% CO
2
in air, 4–10% CO
2
is
common for most cell culture experiments. However, each medium has a recommended
CO
2
tension and bicarbonate concentration to achieve the correct pH and osmolality; refer
to the media manufacturer’s instructions for more information.
Temperature The optimal temperature for cell culture largely depends on the body temperature of
the host from which the cells were isolated, and to a lesser degree on the anatomical
variation in temperature (e.g., temperature of the skin may be lower than the temperature
of skeletal muscle). Overheating is a more serious problem than underheating for cell
cultures; therefore, often the temperature in the incubator is set slightly lower than the
optimal temperature.
• Most human and mammalian cell lines are maintained at 36°C to 37°C for optimal
growth.
• Insect cells are cultured at 27°C for optimal growth; they grow more slowly at lower
temperatures and at temperatures between 27°C and 30°C. Above 30°C, the viability of
insect cells decreases, and the cells do not recover even after they are returned to 27°C.
• Avian cell lines require 38.5°C for maximum growth. Although these cells can also be
maintained at 37°C, they will grow more slowly.
• Cell lines derived from cold-blooded animals (e.g., amphibians, cold-water fish)
tolerate a wide temperature range between 15°C and 26°C.
Note that cell culture conditions vary for each cell type. The consequences of deviating
from the culture conditions required for a particular cell type can range from the
expression of aberrant phenotypes to a complete failure of the cell culture. We therefore
recommend that you familiarize yourself with your cell line of interest, and closely follow
the instructions provided with each product you are using in your experiments.

22 | Cell Culture Basics
Part 3. Basic Cell Culture
Cell Morphology
Regularly examining the morphology of the cells in culture (i.e., their shape and
appearance) is essential for successful cell culture experiments. In addition to confirming
the healthy status of your cells, inspecting the cells by eye and a microscope each time
they are handled will allow you to detect any signs of contamination early on and to
contain it before it spreads to other cultures around the laboratory.
Signs of deterioration of cells include granularity around the nucleus, detachment of
the cells from the substrate, and cytoplasmic vacuolation. Signs of deterioration may be
caused by a variety of reasons, including contamination of the culture, senescence of the
cell line, or the presence of toxic substances in the medium, or they may simply imply that
the culture needs a medium change. Allowing the deterioration to progress too far will
make it irreversible.
Mammalian Cells
Variations in
Mammalian Cell
Morphology
Most mammalian cells in culture can be divided in to three basic categories based on their
morphology.
• Fibroblastic (or fibroblast-like) cells are bipolar or multipolar and have elongated
shapes. They grow attached to a substrate.
• Epithelial-like cells are polygonal in shape with more regular dimensions, and grow
attached to a substrate in discrete patches.
• Lymphoblast-like cells are spherical in shape and they are usually grown in
suspension without attaching to a surface.
In addition to the basic categories listed above, certain cells display morphological
characteristics specific to their specialized role in host.
• Neuronal cells exist in different shapes and sizes, but they can roughly be divided
into two basic morphological categories, type I with long axons used to move signals
over long distances and type II without axons. A typical neuron projects cellular
extensions with many branches from the cell body, which is referred to as a dendritic
tree. Neuronal cells can be unipolar or pseudounipolar with the dendrite and axon
emerging from same process, bipolar with the axon and single dendrite on opposite
ends of the soma (the central part of the cell containing the nucleus), or multipolar with
more than two dendrites.

Cell Culture Basics | 23
Part 3. Basic Cell Culture
Morphology of 293
Cells
The 293 cell line is a permanent line established from primary embryonic human kidney,
which was transformed with sheared human adenovirus type 5 DNA. The adenoviral
genes expressed in this cell line allow the cells to produce very high levels of recombinant
proteins. Invitrogen offers several variants of the 293 cell line, including those adapted
for high-density suspension culture in serum-free media. For more information, visit our
mammalian cell culture pages on our website.
The phase contrast images below show the morphology of healthy 293 cells in adherent
culture at 80% confluency (Figure 3.1) and in suspension culture (Figure 3.2). Note that
adherent mammalian cultures should be passaged when they are in the log phase, before
they reach confluence (see When to Subculture in the Methods chapter, page 27).
A B
Figure 3.1. Phase contrast images of healthy 293 cells in adherent culture. The cells were plated at a seeding density
of 5×10
4
viable cells/cm
2
in 293 SFM II medium and grown as a monolayer in a 37°C incubator with a humidified
atmosphere of 5% CO
2
in air. The images were obtained using 10X and 20X objectives (panels A and B, respectively)
4 days after plating.
A B
Figure 3.2. Phase contrast images of healthy 293F cells grown is suspension. The culture was started in a shake flask
at a seeding density of 2×10
5
viable cells/mL in 293 SFM II medium and grown in a 37°C incubator with a humidified
atmosphere of 5% CO
2
in air. 4days after seeding, the cells were diluted 1:3, and the images were obtained using
10X and 20X objectives (panels A and B, respectively).

24 | Cell Culture Basics
Part 3. Basic Cell Culture
Insect Cells
Morphology of Sf21
Cells
Sf21 cells (IPLB-Sf21-AE) are ovarian cells isolated from Spodoptera frugiperda (Fall
Armyworm). They are spherical in shape with unequal sizes, and have a somewhat
granular appearance. Sf21 cells can be thawed and used directly in suspension culture
for rapid expansion of cell stocks, propagation of baculovirus stocks, and production of
recombinant proteins. Because Sf21 cells attach firmly to surfaces, they can be used as a
monolayer for transfection or plaque assay applications.
The images below show the morphology of healthy Sf21 insect cells in suspension
culture (Figure 3.3) and in adherent culture at confluency (Figure 3.4). Note that insect
cells should be subcultured when they reach confluency (see When to Subculture in the
Methods chapter, page 27).
Figure 3.3. Phase contrast images of healthy Sf21 insect cells grown in suspension. The culture was started in a
shake flask at a seeding density of 3×10
5
viable cells/mL in Sf-900 II SFM medium and it was maintained in a 28°C,
non-humidified, ambient air-regulated incubator. The images were obtained using 10X and 20X objectives (panels
A and B, respectively) 3 days after seeding.
A B
Figure 3.4. Phase contrast images of Sf21 insect cells grown as an adherent monolayer in 293 SFM II medium. The
cells were plated at a seeding density of 5×10
4
viable cells/cm
2
in a T-25 flask and grown as monolayers in a 28°C,
non-humidified, ambient air-regulated incubator. The images were obtained using 10X and 20X objectives (panels
A and B, respectively) 7 days after seeding, when the culture had reached confluency.
A B

Cell Culture Basics | 25
Part 3. Basic Cell Culture
Morphology of Sf9
Cells
The Sf9 insect cell line is a clonal isolate derived from the parental Spodoptera frugiperda
cell line IPLB-Sf-21-AE, and it is a suitable host for expression of recombinant proteins
from baculovirus expression systems (e.g., Invitrogen’s Bac-to-Bac
®
and Bac-N-Blue
™
Expression Systems). Although insect cells have been historically cultured in stationary
systems utilizing T-flasks and serum-supplemented basal medium, insect cells are
generally not anchorage dependent and can easily be maintained in suspension culture.
The images below show the morphology of healthy Sf9 insect cells in suspension and
adherent cultures. Sf9 cells attach firmly to surfaces, and their small, regular size makes
them exceptional for the formation of monolayers and plaques.
Figure 3.5. Phase contrast images of healthy Sf9 insect cells grown in suspension. The culture was started in a
shake flask at a seeding density of 3×10
5
viable cells/mL in Sf-900 II SFM medium and it was maintained in a 28°C,
non-humidified, ambient air-regulated incubator. The images were obtained using 10X and 20X objectives (panels
A and B, respectively) 3 days after seeding.
A B
Figure 3.6. Phase contrast images of healthy Sf9 insect cells grown as an adherent monolayer in 293 SFM II medium.
The cells were plated at a seeding density of 5×10
4
viable cells/cm
2
in a T-25 flask and grown as monolayers in a
28°C, non-humidified, ambient air-regulated incubator. The images were obtained using 10X and 20X objectives
(panels A and B, respectively) 3 days after seeding.
A B

26 | Cell Culture Basics
Methods
Guidelines for Maintaining Cultured Cells
This section provides guidelines and general procedures for routine subculturing,
thawing, and freezing of cells in culture. Note that cell culture conditions vary for each
cell type. The consequences of deviating from the culture conditions required for a
particular cell type can range from the expression of aberrant phenotypes to a complete
failure of the cell culture. We therefore recommend that you familiarize yourself with your
cell line of interest, and closely follow the instructions provided with each product you are
using in your experiments.
What is Subculture? Subculturing, also referred to as passaging, is the removal of the medium and transfer
of cells from a previous culture into fresh growth medium, a procedure that enables the
further propagation of the cell line or cell strain.
The growth of cells in culture proceeds from the lag phase following seeding to the log
phase, where the cells proliferate exponentially. When the cells in adherent cultures
occupy all the available substrate and have no room left for expansion, or when the cells
in suspension cultures exceed the capacity of the medium to support further growth, cell
proliferation is greatly reduced or ceases entirely (see Figure 4.1 below). To keep them at
an optimal density for continued growth and to stimulate further proliferation, the culture
has to divided and fresh medium supplied.
10
4
10
5
10
6
Days
0 8
642 10
Cell density
Subculture
Log phase growth
Figure 4.1. Characteristic Growth Pattern of Cultured Cells. The semi-logarithmic plot shows the cell density versus the
time spent in culture. Cells in culture usually proliferate following a standard growth pattern. The first phase of growth
after the culture is seeded is the lag phase, which is a period of slow growth when the cells are adapting to the culture
environment and preparing for fast growth. The lag phase is followed by the log phase (i.e., “logarithmic” phase), a period
where the cells proliferate exponentially and consume the nutrients in the growth medium. When all the growth medium
is spent (i.e., one or more of the nutrients is depleted) or when the cells occupy all of the available substrate, the cells enter
the stationary phase (i.e., plateau phase), where the proliferation is greatly reduced or ceases entirely.

Cell Culture Basics | 27
Part 4. Methods
When to Subculture? The criteria for determining the need for subculture are similar in adherent and
suspension cultures; however, there are some differences between mammalian and insect
cell lines.
Cell Density
• Mammalian cells: Adherent cultures should be passaged when they are in the log
phase, before they reach confluence. Normal cells stop growing when they reach
confluence (contact inhibition), and it takes them longer to recover when reseeded.
Transformed cells can continue proliferating even after they reach confluence, but they
usually deteriorate after about two doublings. Similarly, cells in suspension should be
passaged when they are in log-phase growth before they reach confluency. When they
reach confluency, cells in suspension clump together and the medium appears turbid
when the culture flask is swirled.
• Insect cells: Insect cells should be subcultured when they are in the log phase,
before they reach confluency. While tightly adherent insect cells can be passaged at
confluency, which allows for easier detachment from the culture vessel, insect cells that
are repeatedly passaged at densities past confluency display decreased doubling times,
decreased viabilities, and a decreased ability to attach. On the other hand, passaging
insect cells in adherent culture before they reach confluency requires more mechanical
force to dislodge them from the monolayer. When repeatedly subcultured before
confluency, these cells also display decreased doubling times and decreased viabilities,
and are considered unhealthy.
Exhaustion of Medium
• Mammalian cells: A drop in the pH of the growth medium usually indicates a build up
of lactic acid, which is a by-product of cellular metabolism. Lactic acid can be toxic to
the cells, and the decreased pH can be sub-optimal for cell growth. The rate of change
of pH is generally dependent on the cell concentration in that cultures at a high cell
concentration exhaust medium faster than cells lower concentrations. You should
subculture your cells if you observe a rapid drop in pH (> 0.1–0.2 pH units) with an
increase in cell concentration.
• Insect cells: Insect cells are cultured in growth media that are usually more acidic that
those used for mammalian cells. For example, TNM-FH and Grace’s medium used
for culturing Sf9 cells has a pH of 6.2. Unlike mammalian cell cultures, the pH rises
gradually as the insect cells grow, but usually does not exceed pH 6.4. However, as
with mammalian cells, the pH of the growth medium will start falling when insect cells
reach higher densities.
Subculture Schedule
Passaging your cells according to a strict schedule ensures reproducible behavior and
allows you to monitor their health status. Vary the seeding density of your cultures until
you achieve consistent growth rate and yield appropriate for your cell type from a given
seeding density. Deviations from the growth patterns thus established usually indicate
that the culture is unhealthy (e.g., deterioration, contamination) or a component of your
culture system is not functioning properly (e.g., temperature is not optimal, culture
medium too old). We strongly recommend that you keep a detailed cell culture log, listing
the feeding and subculture schedules, types of media used, the dissociation procedure
followed, split ratios, morphological observations, seeding concentrations, yields, and any
anti-biotic use.
It is best to perform experiments and other non-routine procedures (e.g., changing type of
media) according to your subculture schedule. If your experimental schedule does not fit
the routine subculture schedule, make sure that you do not passage your cells while they
are still in the lag period or when they have reached confluency and ceased growing.

28 | Cell Culture Basics
Part 4. Methods
Media
Recommendations
Many continuous mammalian cell lines can be maintained on a relatively simple medium
such as MEM supplemented with serum, and a culture grown in MEM can probably be
just as easily grown in DMEM or Medium 199. However, when a specialized function
is expressed, a more complex medium may be required. Information for selecting the
appropriate medium for a given cell type is usually available in published literature, and
may also be obtained from the source of the cells or cell banks.
If there is no information available on the appropriate medium for your cell type, choose
the growth medium and serum empirically or test several different media for best results.
In general, a good place to start is MEM for adherent cells and RPMI-1640 for suspension
cells. The conditions listed below can be used as a guide line when setting up a new
mammalian cell culture.
Insect cells are cultured in growth media that are usually more acidic that those used for
mammalian cells such as TNM-FH and Grace’s medium
Mammalian Cell Culture
Cell Line Cell Type Species Tissue Medium*
293 broblast human embryonic kidney MEM and 10% FBS
3T6 broblast mouse embryo DMEM, 10% FBS
A549 epithelial human lung carcinoma F-12K, 10% FBS
A9 broblast mouse connective tissue DMEM, 10% FBS
AtT-20 epithelial mouse pituitary tumor F-10, 15% horse serum, and 2.5% FBS
BALB/3T3 broblast mouse embryo DMEM, 10% FBS
BHK-21 broblast hamster kidney
GMEM, 10% FBS, or MEM, 10% FBS, and
NEAA
BHL-100 epithelial human breast McCoy'5A, 10% FBS
BT broblast bovine turbinate cells MEM, 10% FBS, and NEAA
Caco-2 epithelial human colon adeno carcinoma MEM, 20% FBS, and NEAA
Chang epithelial human liver BME, 10% calf serum
CHO-K1 epithelial hamster ovary F-12, 10% FBS
Clone 9 epithelial rat liver F-12K, 10% FBS
Clone M-3 epithelial mouse melanoma F-10, 15% horse serum, and 2.5% FBS
COS-1, COS-3, COS-7 broblast monkey kidney DMEM, 10% FBS
CRFK epithelial cat kidney MEM, 10% FBS, and NEAA
CV-1 broblast monkey kidney MEM, 10% FBS
D-17 epithelial dog osteosarcoma MEM, 10% FBS, and NEAA
Daudi lymphoblast human blood from a lymphoma patient RPMI-1640, 10% FBS
GH1, GH3 epithelial rat pituitary tumor F-10, 15% horse serum, and 2.5% FBS
* BME: Basal Medium Eagle; DMEM: Dulbecco’s Modied Eagle Medium; FBS: Fetal Bovine Serum; GMEM: Glasgow Minimum Essential Medium;
IMDM: Iscove’s Modied Dulbecco’s Medium; MEM: Minimum Essential Medium; NEAA: Non-Essential Amino Acids Solution.

Cell Culture Basics | 29
Part 4. Methods
Mammalian Cell Culture, continued
Cell Line Cell Type Species Tissue Medium*
H9 lymphoblast human T-cell lymphoma RPMI-1640, 20% FBS
HaK epithelial hamster kidney BME, 10% calf serum
HCT-15 epithelial human colorectal adenocarcinoma RPMI-1640, 10% FBS
HeLa epithelial human cervix carcinoma MEM, 10% FBS, and NEAA (in suspension, S-MEM)
HEp-2 epithelial human larynx carcinoma MEM, 10% FBS
HL-60 lymphoblast human promyeolocytic leukemia RPMI-1640, 20% FBS
HT-1080 epithelial human brosarcoma MEM, 10% HI FBS, and NEAA
HT-29 epithelial human colon adenocarcinoma McCoy's 5A, 10% FBS
HUVEC endothelial human umbilical cord F-12K, 10% FBS, and 100 μg/mL heparin
I-10 epithelial mouse testicular tumor F-10, 15% horse serum, and 2.5% FBS
IM-9 lymphoblast human marrow from myeloma patient RPMI-1640, 10% FBS
JEG-2 epithelial human choriocarcinoma MEM, 10% FBS
Jensen broblast rat sarcoma McCoy's 5A, 5% FBS
Jurkat lymphoblast human lymphoma RPMI-1640, 10% FBS
K-562 lymphoblast human myelogenous leukemia RPMI-1640, 10% FBS
KB epithelial human oral carcinoma MEM, 10% FBS, and NEAA
KG-1 myeloblast human marrow from erythroleukemia patient IMDM, 20% FBS
L2 epithelial rat lung F-12K, 10%FBS
LLC-WRC 256 epithelial rat carcinoma Medium 199, 5% horse serum
McCoy broblast mouse unknown MEM, 10% FBS
MCF7 epithelial human breast adenocarcinoma MEM, 10% FBS, NEAA, and 10 μg/mL insulin
WI-38 epithelial human embryonic lung BME, 10% FBS
WISH epithelial human amnion BME, 10% FBS
XC epithelial rat sarcoma MEM, 10% FBS, and NEAA
Y-1 epithelial mouse tumor of adrenal F-10, 15% horse serum, and 2.5% FBS
Insect Cell Culture
Sf9, Sf21
fall army worm
(Spodopterafrugiperda)
pupal ovary
TNM-FH and 10% FBS, or Sf-900 II SFM (serum-
free), or Sf-900™ III SFM (serum-free)
High Five™
(BTI-TN-5B1-4)
cabbage looper
(Trichoplusiani)
ovary
TNM-FH and 10% FBS, or Express Five® SFM
(serum-free)
Schneider 2 (S2),
D.Mel-2
fruit y
(Drosophila melanogaster)
Schneider’s Drosophila medium and 10% heat-
inactivated FBS
* BME: Basal Medium Eagle; DMEM: Dulbecco’s Modied Eagle Medium; FBS: Fetal Bovine Serum; GMEM: Glasgow Minimum Essential Medium;
IMDM: Iscove’s Modied Dulbecco’s Medium; MEM: Minimum Essential Medium; NEAA: Non-Essential Amino Acids Solution; TNM-FH:
Trichoplusiani Medium-Formulation Hink (i.e., Grace’s Insect Medium, Supplemented).

30 | Cell Culture Basics
Part 4. Methods
Dissociating
Adherent Cells
The first step in subculturing adherent cells is to detach them from the surface of the
culture vessel by enzymatic or mechanical means. The table below lists the various cell
dissociation procedures.
Procedure Dissociation Agent Applications
Shake-o
Gentle shaking or rocking of
culture vessel, or vigorous
pipetting.
Loosely adherent cells, mitotic cells
Scraping Cell scraper
Cell lines sensitive to proteases; may
damage some cells
Enzymatic
dissociation
Trypsin Strongly adherent cells
Trypsin + collagenase
High density cultures, cultures that
have formed multiple layers, especially
broblasts
Dispase
Detaching epidermal cells as
conuent, intact sheets from the
surface of culture dishes without
dissociating the cells
TrypLE™ dissociation enzyme
Strongly adherent cells; direct
substitute for trypsin; applications that
require animal origin-free reagents
TrypLE
™
Dissociation
Enzymes
TrypLE
™
Express and TrypLE
™
Select are microbially produced cell dissociation enzymes
with similar kinetics and cleavage specificities to trypsin. Although TrypLE
™
enzymes can
directly substitute trypsin in dissociation procedures without a need for protocol changes,
we recommend that you initially optimize the incubation time for dissociation for best
results. Because TrypLE
™
enzymes are recombinant fungal trypsin-like proteases, they are
ideal for applications that require animal origin-free reagents. The table below compares
TrypLE
™
Express and TrypLE
™
Select to trypsin.
TrypLE™ Express and TrypLE™ Select Trypsin
Completely free of animal- and human-derived
components
Porcine- or bovine-derived
Stable at room temperature for at least six
months.
Not stable at room temperature.
Does not require inactivation Requires inactivation with serum or other
inhibitors
Does not require trypsin inactivators Requires trypsin inactivators

Cell Culture Basics | 31
Part 4. Methods
Subculturing Adherent Cells
The following protocol describes a general procedure for subculturing adherent
mammalian cells in culture. Note that the procedure for passaging insect cells differs
from that for mammalian cells on several crucial steps. For more information, refer to
Notes on Subculturing Insect Cells, next page.
For passaging your own cell line, we recommend that you closely follow the
instructions provided with each product you are using in your experiments. The
consequences of deviating from the culture conditions required for a particular cell type
can range from the expression of aberrant phenotypes to a complete failure of the cell
culture.
Materials Needed • Culture vessels containing your adherent cells
• Tissue-culture treated flasks, plates or dishes
• Complete growth medium, pre-warmed to 37°C
• Disposable, sterile 15-mL tubes
• 37°C incubator with humidified atmosphere of 5% CO
2
• Balanced salt solution such as Dulbecco’s Phosphate Buffered Saline (DPBS),
containing no calcium, magnesium, or phenol red
• Dissociation reagent such as trypsin or TrypLE
™
Express, without phenol red
• Reagents and equipment to determine viable and total cell counts such as Countess
®
Automated Cell Counter, Trypan Blue and hemacytometer, or Coulter Counter
®
(Beckman Coulter)
Protocol for
Passaging Adherent
Cells
All solutions and equipment that come in contact with the cells must be sterile. Always
use proper sterile technique and work in a laminar flow hood.
1. Remove and discard the spent cell culture media from the culture vessel.
2. Wash cells using a balanced salt solution without calcium and magnesium
(approximately 2 mL per 10 cm
2
culture surface area). Gently add wash solution to
the side of the vessel opposite the attached cell layer to avoid disturbing the cell layer,
and rock the vessel back and forth several times.
Note: The wash step removes any traces of serum, calcium, and magnesium that
would inhibit the action of the dissociation reagent.
3. Remove and discard the wash solution from the culture vessel
4. Add the pre-warmed dissociation reagent such as trypsin or TrypLE
™
to the side of
the flask; use enough reagent to cover the cell layer (approximately 0.5 mL per 10
cm
2
). Gently rock the container to get complete coverage of the cell layer.
5. Incubate the culture vessel at room temperature for approximately 2 minutes. Note
that the actual incubation time varies with the cell line used.
6. Observe the cells under the microscope for detachment. If cells are less than 90%
detached, increase the incubation time a few more minutes, checking for dissociation
every 30 seconds. You may also tap the vessel to expedite cell detachment.

32 | Cell Culture Basics
Part 4. Methods
7. When ≥ 90% of the cells have detached, tilt the vessel for a minimal length of time
to allow the cells to drain. Add the equivalent of 2 volumes (twice the volume used
for the dissociation reagent) of pre-warmed complete growth medium. Disperse the
medium by pipetting over the cell layer surface several times.
8. Transfer the cells to a 15-mL conical tube and centrifuge then at 200 × g for 5 to 10
minutes. Note that the centrifuge speed and time vary based on the cell type.
9. Resuspend the cell pellet in a minimal volume of pre-warmed complete growth
medium and remove a sample for counting.
10. Determine the total number of cells and percent viability using a hemacytometer,
cell counter and Trypan Blue exclusion, or the Countess
®
Automated Cell Counter. If
necessary, add growth media to the cells to achieve the desired cell concentration and
recount the cells.
Note: We recommend using the Countess
®
Automated Cell Counter to determine
the total number of cells and percent viability. Using the same amount of sample
that you currently use with the hemacytometer, the Countess
®
Automated Cell
Counter takes less than a minute per sample for a typical cell count and is compatible
with a wide variety of eukaryotic cells. For more information on using a traditional
hemacytometer, see Support Protocols, page 41.
11. Dilute cell suspension to the seeding density recommended for the cell line, and
pipet the appropriate volume into new cell culture vessels, and return the cells to the
incubator.
Note: If using culture flasks, loosen the caps before placing them in the incubator to
allow proper gas exchange unless you are using vented flasks with gas-permeable
caps.
Notes on
Subculturing
Adherent Insect Cells
While the general procedure for subculturing insect cells follows the same steps as
mammalian cells, some key requirements of these culture systems are different. For best
results, always follow the instructions provided with each product you are using in your
experiments.
• Passage insect cells at log phase. However, if your insect cells are strongly adherent,
you may passage them at confluency or slightly after when they are starting to pull
away from the bottom of the flask because they will be easier to dislodge.
• Densities lower than 20% confluency inhibit growth. The healthiest cells are those
taken from log phase cultures.
• CO
2
exchange is not recommended for insect cell culture.
• Maintain insect cells at 27°C in a non-humidified environment. Cells can be maintained
at room temperature on the bench top if protected from light or in a drawer. However,
a 27°C controlled environment is recommended.
• Use media specifically formulated for insect cell growth.
• Insect cells attach very tightly to substrates under serum-free conditions and require
additional effort to detach. To dislodge the cells, you may need to give the flask one
quick shake using a wrist-snapping motion. To avoid contamination, always tighten
the cap before this procedure.
Caution: We do not recommend shaking the flask vigorously, because it may result in
damage to the cells.

Cell Culture Basics | 33
Part 4. Methods
Subculturing Suspension Cells
The following protocols describe general procedures for subculturing mammalian cells
in suspension culture. Note that the procedure for passaging insect cells differs from
that for mammalian cells on several crucial steps. For more information, refer to Notes on
Subculturing Insect Cells, page 36.
For passaging your own cell line, we recommend that you closely follow the instructions
provided with each product you are using in your experiments. The consequences of
deviating from the culture conditions required for a particular cell type can range from the
expression of aberrant phenotypes to a complete failure of the cell culture.
Passaging
Suspension Cultures
Subculturing suspension cells is somewhat less complicated than passaging adherent
cells. Because the cells are already suspended in growth medium, there is no need to
treat them enzymatically to detach them from the surface of the culture vessel, and the
whole process is faster and less traumatic for the cells. Replacement of growth medium is
not carried out in suspension cultures; instead, the cells are maintained by feeding them
every 2 to 3 days until they reach confluency. This can be done by directly diluting the
cells in the culture flask and continue expanding them, or by withdrawing a portion of
the cells from the culture flask and diluting the remaining cells down to a seeding density
appropriate for the cell line. Usually, the lag period following the passaging is shorter
than that observed with adherent cultures.
Suspension Culture
Vessels
Suspension cultures can be maintained in sterile culture flasks (e.g., shaker flasks without
baffles) that are not tissue-culture treated; however, spinner flasks (i.e., stirrer bottles)
specifically designed for suspension cell culture allow for superior gas exchange and
permit higher volumes of cells to be cultured.
Spinner flasks have two basic designs; the medium is agitated (i.e., stirred) by a hanging
stir-bar assembly or with a vertical impeller. The vertical impeller provides better aeration.
The total culture volume in a spinner flask should not exceed half of the indicated volume
of the spinner for proper aeration (e.g., a 500 mL spinner should never contain more than
250 mL of culture).

34 | Cell Culture Basics
Part 4. Methods
Materials Needed • Culture vessels containing your suspension cells
• Shaker flasks without baffles or spinner bottles (see Suspension Culture Vessels,
previous page)
• Complete growth medium, pre-warmed to 37°C
• 37°C incubator with humidified atmosphere of 5% CO
2
• Magnetic stir plate (if using spinner flasks), roller rack (if using roller bottles), or
shaking platform (if using conventional culture flasks or petri dishes)
• Reagents and equipment to determine viable and total cell counts (e.g., Countess
®
Automated Cell Counter, Trypan Blue and hemacytometer, or Coulter Counter
®
)
Protocol for
Passaging
Suspension Cells
All solutions and equipment that come in contact with the cells must be sterile. Always
use proper sterile technique and work in a laminar flow hood. Subculture cells when they
are in log-phase growth before they reach confluency. When they reach confluency, cells
in suspension clump together and the medium appears turbid when the culture flask is
swirled. The maximum recommended cell density before passaging varies with cell lines;
refer to the cell-specific product insert or manual for details.
Cells Grown in Shaker Flasks
The following protocol describes a general procedure for passaging mammalian cells
grown in suspension culture using shaker flasks in a shaking incubator. For detailed
protocols, always refer to the cell-specific product insert.
Note: Make sure that the shaker flask does not have baffles (i.e., the indents at the bottom
of the flask designed to provide agitation), because they ruin the shaking rhythm.
1. When the cells are ready for passaging (i.e., log-phase growth before they reach
confluency), remove the flask from the incubator and take a small sample from
the culture flask using a sterile pipette. If cells have settled down before taking the
sample, swirl the flask to evenly distribute the cells in the medium.
2. From the sample, determine the total number of cells and percent viability using the
Countess
®
Automated Cell Counter or a hemacytometer, cell counter, and Trypan
Blue exclusion.
3. Calculate the volume of media that you need to add to dilute the culture down to the
recommended seeding density.
4. Aseptically add the appropriate volume of pre-warmed growth medium into the
culture flask. You may split the culture to multiple flasks if needed.
5. Loosen the caps of the culture flasks one full turn to allow for proper gas exchange (or
use a gas-permeable cap), and return the flasks to the shaking incubator. The shaking
speed depends on the cell line.
Note: To minimize the accumulation of cell debris and metabolic waste by-products in
shaker cultures, gently centrifuge the cell suspension at 100 × g for 5 to 10 minutes, and
resuspend the cell pellet in fresh growth medium once every three weeks (or as needed).

Cell Culture Basics | 35
Part 4. Methods
Cells Grown in Spinner Flasks
The following protocol describes a general procedure for passaging mammalian cells in
suspension grown using spinner flasks. For detailed protocols, always refer to the cell-
specific product insert.
Note that cells are sensitive to physical shearing. Ensure that impeller mechanisms rotate
freely and do not contact vessel walls or the base. The top of the paddles should be
slightly above the medium to ensure adequate aeration to the culture. Adjust the spinner
mechanism so that paddles clear the sides and the bottom of the vessel. The table below
lists the minimum volumes of media needed for different spinner flask sizes.
We do not recommend initiating a spinner culture into a spinner flask larger than 500mL.
We suggest scaling up from smaller spinners that have already been established.
1. When the cells are ready for passaging (i.e., log-phase growth before they reach
confluency), remove the flask from the incubator, and take a small sample from
the culture flask using a sterile pipette. If cells have settled down before taking the
sample, swirl the to evenly distribute the cells in the medium.
2. From the sample, determine the total number of cells and percent viability using the
Countess
®
Automated Cell Counter or a hemacytometer, cell counter and Trypan Blue
exclusion.
3. Calculate the volume of media that you need to add to dilute the culture down to the
recommended seeding density.
4. Aseptically add the appropriate volume of pre-warmed growth medium into the
culture flask. You may split the culture to multiple flasks if needed.
5. Loosen the side arm caps of the spinner flasks one full turn to allow for proper gas
exchange, and return the flasks to the incubator. The spinner speed depends on the
cell line and the impeller type. Make sure that the spinner speed is kept within the
recommended values to avoid damage to the cells from shear stress.
Note: To minimize the accumulation of cell debris and metabolic waste by-products in
spinner cultures, gently centrifuge the cell suspension at 100 × g for 5 to 10 minutes, and
resuspend the cell pellet in fresh growth medium once every three weeks (or as needed).
Size of Spinner Flask Minimum Media Volume
100 mL 30 mL
250 mL 80 mL
500 mL 200 mL

36 | Cell Culture Basics
Part 4. Methods
Notes on
Subculturing
Suspension Insect
Cells
While the general procedure for subculturing insect cells follows the same steps as
mammalian cells, some key requirements of these culture systems are different. For best
results, always follow the instructions provided with the insect cell lines you are using
in your experiments.
• It is not necessary to change medium when you are culturing cells in suspension.
Regular subculturing requires the removal of cell suspension and the addition of
medium sufficient to dilute culture to the appropriate density (refer to the cell-specific
product insert). Adding fresh medium is sufficient to replenish cell nutrients.
• CO
2
exchange is not recommended for insect cell culture.
• Maintain insect cells at 27°C in a non-humidified environment. Cells can be maintained
at room temperature on the bench top or in a drawer, however, a 27°C controlled
environment is recommended.
• Use media specifically formulated for insect cell growth.
• Use a surfactant to decrease shearing. 0.1% Pluronic
®
F-68 is recommended for spinner
insect cultures. Pluronic
®
F-68 (BASF) is a surfactant that decreases cell membrane
shearing due to impeller forces.
Note: Sf-900 II SFM and Express Five
®
SFM already contain surfactants.
• Certain insect cell lines may require adaptation to suspension culture. For more
information, refer to the cell-line specific product insert or manual.

Cell Culture Basics | 37
Part 4. Methods
Freezing Cells
Cryopreservation Cell lines in continuous culture are prone to genetic drift, finite cell lines are fated for
senescence, all cell cultures are susceptible to microbial contamination, and even the
best-run laboratories can experience equipment failure. Because an established cell line
is a valuable resource and its replacement is expensive and time consuming, it is vitally
important that they are frozen down and preserved for long-term storage.
As soon as a small surplus of cells becomes available from subculturing, they should be
frozen as a seed stock, protected, and not be made available for general laboratory use.
Working stocks can be prepared and replenished from frozen seed stocks. If the seed
stocks become depleted, cryopreserved working stocks can then serve as a source for
preparing a fresh seed stock with a minimum increase in generation number from the
initial freezing.
The best method for cryopreserving cultured cells is storing them in liquid nitrogen in
complete medium in the presence of a cryoprotective agent such as dimethylsulfoxide
(DMSO). Cryoprotective agents reduce the freezing point of the medium and also allow a
slower cooling rate, greatly reducing the risk of ice crystal formation, which can damage
cells and cause cell death.
Note: DMSO is known to facilitate the entry of organic molecules into tissues. Handle
reagents containing DMSO using equipment and practices appropriate for the hazards
posed by such materials. Dispose of the reagents in compliance with local regulations.
Guidelines for
Cryopreservation
Following the guidelines below is essential for cryopreserving your cell lines for future
use. As with other cell culture procedures, we recommend that you closely follow the
instructions provided with your cell line for best results.
• Freeze your cultured cells at a high concentration and at as low a passage number as
possible. Make sure that the cells are at least 90% viable before freezing. Note that the
optimal freezing conditions depend on the cell line in use.
• Freeze the cells slowly by reducing the temperature at approximately 1°C per minute
using a controlled rate cryo-freezer or a cryo-freezing container such as “Mr. Frosty,”
available from NALGENE
®
labware (Nalge Nunc)
• Always use the recommended freezing medium. The freezing medium should contain
a cryoprotective agent such as DMSO or glycerol (see What is Subculture?, page 26).
• Store the frozen cells below –70°C; frozen cells begin to deteriorate above –50°C.
• Always use sterile cryovials for storing frozen cells. Cryovials containing the frozen
cells may be stored immersed in liquid nitrogen or in the gas phase above the liquid
nitrogen (see Safety Note, page 37).
• Always wear personal protective equipment.
• All solutions and equipment that come in contact with the cells must be sterile. Always
use proper sterile technique and work in a laminar flow hood.
Safety Note
Biohazardous materials must be stored in the gas phase above the liquid nitrogen.
Storing the sealed cryovials in the gas phase eliminates the risk of explosion. If you are
using liquid-phase storage, be aware of the explosion hazard with both glass and plastic
cryovials and always wear a face shield or goggles.

38 | Cell Culture Basics
Part 4. Methods
Freezing Medium Always use the recommended freezing medium for cryopreserving your cells. The
freezing medium should contain a cryoprotective agent such as DMSO or glycerol.
You may also use a specially formulated complete cryopreservation medium such as
Recovery
™
Cell Culture Freezing Medium or Synth-a-Freeze
®
Cryopreservation Medium.
• Recovery
™
Cell Culture Freezing Medium is a ready-to-use complete cryopreservation
medium for mammalian cell cultures, containing an optimized ratio of fetal bovine
serum to bovine serum for improved cell viability and cell recovery after thawing.
• Synth-a-Freeze
®
Cryopreservation Medium is a chemically defined, protein-
free, sterile cryopreservation medium containing 10% DMSO that is suitable for
the cryopreservation of many stem and primary cell types with the exception of
melanocytes.
Materials Needed • Culture vessels containing cultured cells in log-phase of growth
• Complete growth medium
• Cryoprotective agent such as DMSO (use a bottle set aside for cell culture; open only in
a laminar flow hood) or a freezing medium such as Synth-a-Freeze
®
Cryopreservation
Medium or Recovery
™
Cell Culture Freezing Medium
• Disposable, sterile 15-mL or 50-mL conical tubes
• Reagents and equipment to determine viable and total cell counts (e.g., Countess
®
Automated Cell Counter, or hemacytometer, cell counter and Trypan Blue)
• Sterile cryogenic storage vials (i.e., cryovials)
• Controlled rate freezing apparatus or isopropanol chamber
• Liquid nitrogen storage container
For freezing adherent cells, in addition to the above materials, you need:
• Balanced salt solution such as Dulbecco’s Phosphate Buffered Saline (D-PBS),
containing no calcium, magnesium, or phenol red
• Dissociation reagent such as trypsin or TrypLE
™
Express, without phenol red

Cell Culture Basics | 39
Part 4. Methods
Protocol for
Cryopreserving
Cultured Cells
The following protocol describes a general procedure for cryopreserving cultured cells.
For detailed protocols, always refer to the cell-specific product insert.
1. Prepare freezing medium and store at 2° to 8°C until use. Note that the appropriate
freezing medium depends on the cell line.
2. For adherent cells, gently detach cells from the tissue culture vessel following the
procedure used during the subculture. Resuspend the cells in complete medium
required for that cell type.
3. Determine the total number of cells and percent viability using a hemacytometer,
cell counter and Trypan Blue exclusion, or the Countess
®
Automated Cell Counter.
According to the desired viable cell density, calculate the required volume of freezing
medium.
4. Centrifuge the cell suspension at approximately 100–200 × g for 5 to 10 minutes
Aseptically decant supernatant without disturbing the cell pellet.
Note: Centrifugation speed and duration varies depending on the cell type.
5. Resuspend the cell pellet in cold freezing medium at the recommended viable cell
density for the specific cell type.
6. Dispense aliquots of the cell suspension into cryogenic storage vials. As you aliquot
them, frequently and gently mix the cells to maintain a homogeneous cell suspension.
7. Freeze the cells in a controlled rate freezing apparatus, decreasing the temperature
approximately 1°C per minute. Alternatively, place the cryovials containing the cells
in an isopropanol chamber and store them at –80°C overnight.
8. Transfer frozen cells to liquid nitrogen, and store them in the gas phase above the
liquid nitrogen.

40 | Cell Culture Basics
Part 4. Methods
Thawing Frozen Cells
Guidelines for
Thawing
The thawing procedure is stressful to frozen cells, and using good technique and working
quickly ensures that a high proportion of the cells survive the procedure. As with other
cell culture procedures, we recommend that you closely follow the instructions provided
with your cells and other reagents for best results.
• Thaw frozen cells rapidly (< 1 minute) in a 37°C water bath.
• Dilute the thawed cells slowly, using pre-warmed growth medium.
• Plate thawed cells at high density to optimize recovery.
• Always use proper aseptic technique and work in a laminar flow hood.
• Always wear personal protective equipment, including a face mask or goggles.
Cryovials stored in liquid-phase present a risk of explosion when thawed.
• Some freezing media contain DMSO, which is known to facilitate the entry of organic
molecules into tissues. Handle reagents containing DMSO using equipment and
practices appropriate for the hazards posed by such materials.
Materials Needed • Cryovial containing frozen cells
• Complete growth medium, pre-warmed to 37°C
• Disposable, sterile centrifuge tubes
• Water bath at 37°C
• 70% ethanol
• Tissue-culture treated flasks, plates, or dishes
Protocol for Thawing
Frozen Cells
The following protocol describes a general procedure for thawing cryopreserved cells.
For detailed protocols, always refer to the cell-specific product insert.
1. Remove the cryovial containing the frozen cells from liquid nitrogen storage and
immediately place it into a 37°C water bath.
2. Quickly thaw the cells (< 1 minute) by gently swirling the vial in the 37°C water bath
until there is just a small bit of ice left in the vial.
3. Transfer the vial it into a laminar flow hood. Before opening, wipe the outside of the
vial with 70% ethanol.
4. Transfer the thawed cells dropwise into the centrifuge tube containing the desired
amount of pre-warmed complete growth medium appropriate for your cell line.
5. Centrifuge the cell suspension at approximately 200 × g for 5–10 minutes. The actual
centrifugation speed and duration varies depending on the cell type.
6. After the centrifugation, check the clarity of supernatant and visibility of a complete
pellet. Aseptically decant the supernatant without disturbing the cell pellet.
7. Gently resuspend the cells in complete growth medium, and transfer them into the
appropriate culture vessel and into the recommended culture environment.
Note: The appropriate flask size depends on the number of cells frozen in the
cryovial, and the culture environment varies based on the cell and media type.
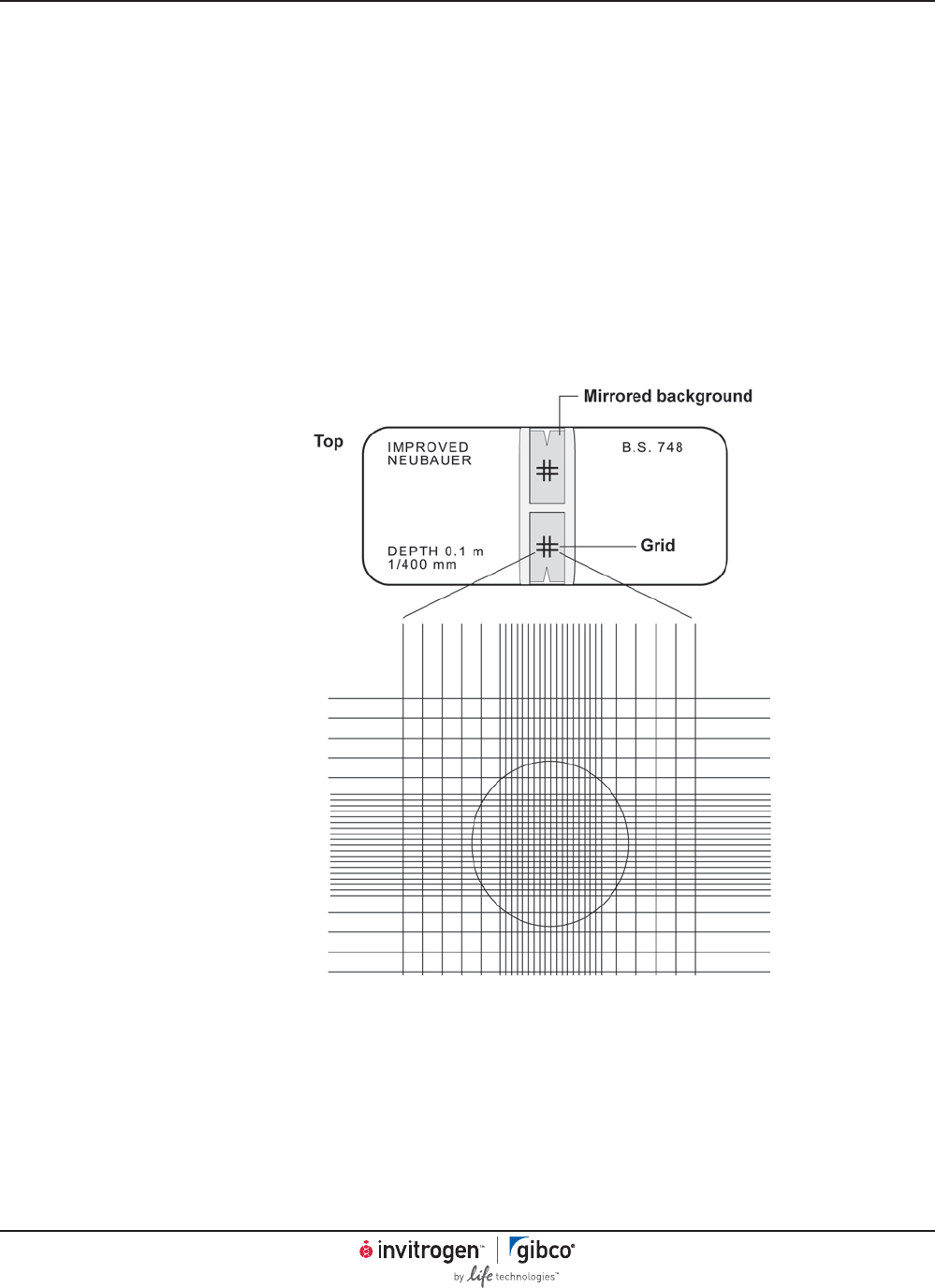
Cell Culture Basics | 41
Part 4. Methods
Support Protocols
Counting Cells in a
Hemacytometer
Hemacytometers may be obtained from most major laboratory suppliers (e.g., Baxter
Scientific). The procedure below provides some general directions on how to use the
hemacytometer.
1. Clean the chamber and cover slip with alcohol. Dry and fix the coverslip in position.
2. Harvest the cells. Add 10 μL of the cells to the hemacytometer. Do not overfill.
3. Place the chamber in the inverted microscope under a 10X objective. Use phase
contrast to distinguish the cells.
4. Count the cells in the large, central gridded square (1 mm
2
). The gridded square is
circled in the graphic below. Multiply by 10
4
to estimate the number of cells per mL.
Prepare duplicate samples and average the count.

42 | Cell Culture Basics
Part 4. Methods
Trypan Blue
Exclusion
The following procedure will enable you to accurately determine the cell viability. Cell
viability is calculated as the number of viable cells divided by the total number of cells
within the grids on the hemacytometer. If cells take up trypan blue, they are considered
non-viable.
1. Determine the cell density of your cell line suspension using a hemacytometer.
2. Prepare a 0.4% solution of trypan blue in buffered isotonic salt solution, pH 7.2 to 7.3
(i.e., phosphate-buffered saline).
3. Add 0.1 mL of trypan blue stock solution to 1 mL of cells.
4. Load a hemacytometer and examine immediately under a microscope at low
magnification.
5. Count the number of blue staining cells and the number of total cells. Cell viability
should be at least 95% for healthy log-phase cultures.
% viable cells = [1.00 – (Number of blue cells ÷ Number of total cells)] × 100
To calculate the number of viable cells per mL of culture, use the formula below.
Remember to correct for the dilution factor.
Number of viable cells × 10
4
× 1.1 = cells/mL culture
Concentrating Cells To concentrate cells from a suspension culture (or resuspended cells from monolayer
culture):
1. Transfer the cell suspension to a sterile centrifuge tube of appropriate size and
centrifuge for 10 minutes at 800 × g.
Note: Certain cell lines are very sensitive to centrifugal force.
2. Carefully remove the supernatant without disturbing the cell pellet.
3. Add the desired volume of fresh medium gently to the side of the tube and slowly
pipette up and down 2 to 3 times to resuspend the cell pellet.
4. Transfer the cells to the desired, sterile container.

Cell Culture Basics | 43
Appendix
Troubleshooting
The table below lists some potential problems and possible solutions that may help you
troubleshoot your cell culture experiments. Note that the list below includes only the most
commonly encountered problems in cell culture, and provides guidelines to solutions
only. To help evaluate your results more successfully, we recommend that you consult
the manuals and product information sheets provided with the products you are using as
well as the published literature and books on the subject.
Problem Reason Solution
No viable cells after thawing
stock
Cells were stored incorrectly Obtain new stock and store in liquid nitrogen. Keep the cells in liquid
nitrogen until thawing.
Home made freezer stock is not
viable
Freeze cells at a density recommended by the supplier.
Use low-passage cells to make your own freezer stocks.
Follow procedures for freezing cells exactly as recommended by
the supplier. Note that the freezing procedure recommended in this
handbook is a general procedure provided as a guideline only.
Obtain new stock.
Cells were thawed incorrectly Follow procedures for thawing cells exactly as recommended by
the supplier. Note that the thawing procedure recommended in this
handbook is a general procedure provided as a guideline only.
Make sure that you thaw the frozen cells quickly, but dilute them
slowly using pre-warmed growth medium before plating.
Thawing medium is not correct Use the medium recommended by the supplier. Make sure the me-
dium is pre-warmed.
Cells are too dilute Plate thawed cells at high density as recommended by the supplier to
optimize recovery.
Cells not handled gently Freezing and thawing procedures are stressful to most cells. Do not
vortex, bang the asks to dislodge the cells (except when culturing
insect cells), or centrifuge the cells at high speeds.
Glycerol used in the freezing
medium was stored in light (if
applicable)
If stored in light, glycerol is gets converted to acrolein, which toxic to
cells. Obtain new stock.
Cells grow slowly Growth medium is not correct Use pre-warmed growth medium as recommended by the supplier.
Serum in the growth medium is of
poor quality
Use serum from a dierent lot.
Cells have been passaged too
many times
Use healthy, low passage-number cells.
Cells were allowed to grow
beyond conuency
Passage mammalian cells when they are in the log-phase before they
reach conuence.
Culture is contaminated with
mycoplasma
Discard cells, media, and reagents. Obtain new stock of cells, and use
them with fresh media and reagents.

44 | Cell Culture Basics
Part 5. Appendix
Cell Culture Products
Life Technologies offers a variety of primary cultures and established cell lines, as well
as reagents, media, sera, and growth factors for your cell culture experiments. The tables
below contain lists of the more commonly used cell lines and other cell culture products
available from Invitrogen, part of Life Technologies. For more information on Invitrogen
and GIBCO
®
products, refer to www.invitrogen.com/cellculture.
Cell Lines In addition to the mammalian and insect cell lines listed below, Invitrogen offers primary
mammalian cells and complete cell culture systems, including keratinocyte, fibroblast,
melanocyte, hepatocyte, corneal and mammary epithelial, large vessel and microvascular
endothelial, smooth muscle, and neuronal cell culture systems. For a comprehensive list of
cells, technical resources and related technologies, visit
www.invitrogen.com/cellculture.
Product Quantity Cat. no.
Mammalian Cell Lines
293-F Cells, SFM adapted (7.5 × 10
6
cells) 1.5 mL 11625-019
293-H Cells, SFM adapted (7.5 × 10
6
cells) 1.5 mL 11631-017
293FT Cell Line 3 × 10
6
cells R700-07
293A Cell Line 3 × 10
6
cells R705-07
GripTite™ 293 MSR Cell line 1 kit R795-07
CHO DG44 Cells (cGMP banked) and Media Kit 1 kit A11000-01
Insect Cell Lines
Sf9 Cells, adapted in Sf-900™ II SFM (1.5 × 10
7
cells) 1.5 mL 11496-015
Sf21 Cells, adapted in Sf-900™ II SFM (1.5 × 10
7
cells) 1.5 mL 11497-013
Mimic™ Sf9 Insect Cells (1 × 10
7
cells) 1 mL 12552-014
Sf9 Cells adapted in Sf-900™ III SFM (1.5 × 10
7
cells) 1 vial 12659-017
Sf21 Cells adapted in Sf-900™ III SFM (1.5 × 10
7
cells) 1 vial 12682-019
Sf9 Frozen Cells (Grace’s media) (1 × 10
7
cells) 1 mL B825-01
Sf21 Frozen Cells (Grace’s media) (1 × 10
7
cells) 1 mL B821-01
High Five™ Cells, adapted to Express Five® SFM (3 × 10
6
cells) 1 mL B855-02

Cell Culture Basics | 45
Part 5. Appendix
Media for
Mammalian Cell
Culture
Life Technologies provides you with all of your cell culture needs through its GIBCO
®
Cell Culture Media, and offers products to support the growth of a range of mammalian
cell lines. All cell culture media products available from Life Technologies are tested
for contamination, and guaranteed for their quality, safety, consistency, and regulatory
compliance. In addition to the media listed below, Life Technologies offers a large
selection of serum-free and specialized media for culturing primary cells, established
cell lines, and stem cells, as well as for virus production, protein expression, stem cell
differentiation, and cytogenetics. For more information and a complete list of cell culture
media, visit www.invitrogen.com/cellculture.
Product* Quantity
†
Cat. no.
D-MEM (1X), liquid (high glucose with no glutamine) 500 mL 31053-028
D-MEM (1X), liquid (low glucose with no glutamine) 500 mL 11880-028
D-MEM (1X), liquid (high glucose with GlutaMAX™-I) 10 × 500 mL 32430-100
D-MEM (1X), liquid (low glucose with GlutaMAX™-I) 10 × 500 mL 21885-108
Advanced D-MEM (1X), liquid (high glucose with no glutamine) 10 × 500 mL 12491-023
D-MEM/F-12, liquid, 1:1 (with GlutaMAX™-I) 10 × 500 mL 31331-093
Advanced D-MEM/F-12, liquid, 1:1 (with no glutamine) 10 × 500 mL 12634-028
Minimum Essential Medium (MEM) (1X), liquid (with no
glutamine)
10 × 500 mL 21090-055
Minimum Essential Medium (MEM) (1X), liquid (with GlutaMAX™-I) 10 × 500 mL 41090-093
Advanced MEM (Minimum Essential Medium) (1X), liquid (with no
glutamine)
10 × 500 mL 12492-021
RPMI Medium 1640 (1X), liquid (with no glutamine) 500 mL 31870-025
RPMI Medium 1640 (1X), liquid (with GlutaMAX™-I) 10 × 500 mL 61870-044
Advanced RPMI Medium 1640 (1X), liquid (with no glutamine) 10 × 500 mL 12633-020
293 SFM II, liquid 1,000 mL 11686-029
CD 293 Medium, liquid 1,000 mL 11913-019
GIBCO® Freestyle™ 293 Expression Medium 1,000 mL 12338-018
CD CHO Medium (1X), liquid 1,000 mL 10743-029
CHO-S-SFM II 1,000 mL 12052-098
GIBCO® Freestyle™ CHO Expression Medium 1,000 mL 12651-014
CD OptiCHO™ Medium (1X), liquid 1,000 mL 12681-011
Recovery™ Cell Culture Freezing Medium, liquid 50 mL 21530-027
Synth-a-Freeze® Cryopreservation Medium 50 mL R-005-50
*
Most of the media listed in this table are available with L-glutamine, GlutaMAX™-I, or no glutamine, with or
without phenol red, as well as in powder and liquid formulations. †Also available in different quantities and
packaging sizes.

46 | Cell Culture Basics
Part 5. Appendix
Media for Insect Cell
Culture
Insect cell culture is a common choice for heterologous protein expression. For large
scale production or basic research, insect cells are able to express large quantities of
protein with complex post-translational modifications. GIBCO
®
insect media from Life
Technologies have been formulated for maximum growth and protein yields. For more
information, visit www.invitrogen.com.
Product* Quantity
†
Cat. no.
Grace’s Insect Cell Medium, Unsupplemented (1X), liquid 500 mL 11595-030
Grace’s Insect Cell Medium, Supplemented (1X), liquid 500 mL 11605-045
Sf-900™ II SFM (1X), liquid 500 mL 10902-096
Sf-900™ III SFM (1X), liquid 500 mL 12658-019
Schneider’s Drosophila Medium (1X), liquid 500 mL 21720-024
IPL-41 Insect Medium (1X), liquid 500 mL 11405-057
Express Five® SFM (1X), liquid 1,000 mL 10486-025
*
Most of the media listed in this table are available in powder and liquid formulations. †Also available in
different quantities and packaging sizes.

Cell Culture Basics | 47
Part 5. Appendix
Serum Products
for Cell Culture
Life Technologies supplies a wide range of GIBCO
®
animal sera, both bovine and non-
bovine, for cell culture applications, the most widely used being fetal bovine serum
(FBS). The table below lists a small selection of sera available from Life Technologies.
For a complete list and more information on the use, sources, traceability, collection, and
bottling of serum, refer to www.invitrogen.com.
Product Source Quantity Cat. no.
Certied FBS United States 100 mL
500 mL
16000-036
16000-044
Certied,
heat-inactivated FBS
United States 100 mL
500 mL
10082-139
10082-147
Qualied FBS United States 100 mL
500 mL
26140-087
26140-079
Australia 100 mL
500 mL
10099-133
10099-141
Countries that meet EU importation
requirements (South America)
100 mL
500 mL
10270-098
10270-106
Countries that meet EU importation
requirements (excluding South America)
100 mL
500 mL
10106-151
10106-169
Qualied,
heat-inactivated FBS
United States 100 mL
500 mL
16140-063
16140-071
Australia 100 mL
500 mL
10100-139
10100-147
Countries that meet EU importation
requirements (South America)
100 mL
500 mL
10500-056
10500-064
Countries that meet EU importation
requirements (excluding South America)
100 mL
500 mL
10108-157
10108-165
Qualied, gamma-
irradiated FBS
Countries that meet EU importation
requirements (including South America)
100 mL
500 mL
10499-036
10499-044
Countries that meet EU importation
requirements (excluding South America)
100 mL
500 mL
10109-155
10109-165
Horse Serum
New Zealand 100 mL
500 mL
16050-130
16050-122
Horse Serum,
heat-inactivated
New Zealand 100 mL
500 mL
26050-070
26050-088
Porcine Serum
New Zealand 500 mL
26250-084
Rabbit Serum
United States 100 mL
500 mL
16120-099
16120-107
Goat Serum
New Zealand 100 mL
500 mL
16210-064
16210-072
Chicken Serum
New Zealand 500 mL
16110-082
Lamb Serum
New Zealand 500 mL
16070-096
Bovine Serum
New Zealand 100 mL
500 mL
16170-086
16170-078

48 | Cell Culture Basics
Part 5. Appendix
Laboratory Reagents
for Cell Culture
The table below lists a small sampling of laboratory reagents for cell culture that are
available from Life Technologies. For more information and a complete list, refer to
www.invitrogen.com.
Product Quantity* Cat. no.
Balanced Salt Solutions: D-PBS
†
, EBSS, HBSS
‡
, PBS
Dulbecco’s Phosphate Buered Saline (D-PBS) (1X), liquid 1,000 mL 14190-136
Earle’s Balanced Salt Solution (EBSS) (1X), liquid 500 mL 14155-063
Hank’s Balanced Salt Solution (HBSS) (1X), liquid 1,000 mL 14025-076
Phosphate-Buered Saline (PBS) pH 7.4 (1X), liquid 500 mL 10010-023
Phosphate-Buered Saline (PBS) pH 7.2 (1X), liquid 500 mL 70013-032
Buers and Chemicals
HEPES Buer Solution (1M) 20 × 100 mL 15630-130
Sodium Bicarbonate Solution, 7.5% (w/v) 100 mL 25080-094
Cell Dissociation Reagents
TrypLE™ Express Dissociation Reagent with Phenol Red 500 mL 12605-028
TrypLE™ Express Dissociation Reagent without Phenol Red 100 mL 12604-013
TrypLE™ Select Dissociation Reagent 500 mL 12563-029
Trypsin, 0.5% (10X), liquid, with EDTA 4Na, without Phenol Red 100 mL 15400-054
Trypsin, 0.25% (10X), liquid, without EDTA, with Phenol Red 500 mL 15050-057
Collagenase Type I 1 g 17100-017
Collagenase Type II 1 g 17101-015
Dispase 5 g 17105-041
Trypsin Inhibitor, soybean 1 g 17075-029
Supplements
L-Glutamine
†
, 200 mM (100X), liquid 100 mL 25030-081
GlutaMAX™-I Supplement 100 mL 35050-061
D-Glucose (Dextrose) 1 kg 15023-021
Pluronic® F-68, 10% (100X) 100 mL 24040-032
MEM Amino Acids Solution (50X), liquid 100 mL 11130-051
MEM Non-Essential Amino Acids Solution 10 mM (100X), liquid 100 mL 11140-050
MEM Sodium Pyruvate Solution 10 mM (100X), liquid 100 mL 11360-070
MEM Vitamin Solution (100X), liquid 100 mL 11120-052
2-Mercaptoethanol (1,000X), liquid 50 mL 21985-023
*
Products are also available in different quantities and packaging sizes. †Product is available in liquid or powder
formats. ‡HBSS is available with or without magnesium and calcium, and with or without phenol red.

Cell Culture Basics | 49
Part 5. Appendix
Product Quantity* Cat. no.
Antibiotics and Antimycotics
Antibiotic-Antimycotic (100X), liquid 100 mL 15240-062
Fungizone® Antimycotic, liquid 20 mL 15290-018
Gentamycin Reagent Solution (10 mg/mL), liquid 10 mL 15710-064
Gentamycin Reagent Solution (50 mg/mL), liquid 10 mL 15750-060
Gentamycin/Amphotericin Solution 10 × 1 mL R-015-10
Neomycin Sulfate, powder 100 g 21810-031
Penicillin-Streptomycin, liquid 100 mL 15140-122
Penicillin-Streptomycin-Neomycin (PSN) Antibiotic Mixture 100 mL 15640-055
Selection Antibiotics
Geneticin® Selective Antibiotic, liquid 20 mL 10131-027
Geneticin® Selective Antibiotic, powder 1 g 11811-023
Hygromycin B 20 mL 10687-010
Puromycin Dihydrochloride, Selection Antibiotic, liquid 10 × 1 mL A11138-03
Blasticidin S HCl, Selection Antibiotic, liquid 20 mL A11139-02
Blasticidin S HCl, powder 50 mg R210-01
Zeocin™ Selection Reagent, powder 1 g R250-01
Contamination Detection Kits
Cell Culture Contamination Detection Kit - 200 assays 1 kit C7028
MycoFluor™ Mycoplasma Detection Kit 1 kit M7006
*
Products are also available in different quantities and packaging sizes.
Antibiotics and
Antimycotics
Antibiotics are used to protect the integrity of your cell culture as well for selection
and establishment of cell lines; Life Technologies offers a wide selection of antibiotics,
antimycotics and detection kits. For more information, refer to www.invitrogen.com.

50 | Cell Culture Basics
Part 5. Appendix
Accessory Products
for Cell Culture
The table below lists a small sampling of accessory products for cell culture that are
available from Life Technologies. For more information and a complete list, refer to
www.invitrogen.com.
Product Quantity* Cat. no.
Attachment Factors and Matrices
Geltrex™ Reduced Growth Factor Basement Membrane Matrix 5 mL 12760-021
Geltrex™ LDEV-free Reduced Growth Factor Basement
Membrane Matrix Extract
5 mL
A11343-01
Geltrex™ hESC-qualied Reduced Growth Factor Basement
Membrane Matrix
1 mL
5 mL
A10480-01
A10480-02
AlgiMatrix™ 3D culture System 6-well plates
1 plate
A10982-01
AlgiMatrix™ 3D culture System 24-well plates
1 plate
12684-023
AlgiMatrix™ 3D culture System 96-well plates
1 plate
12684-015
Human Plasma Fibronectin
5 mg
33016-015
Natural Mouse Laminin
1 mg
23017-015
Collagen I, Rat Tail
20 mL
A1048301
Collagen I, Bovine
10 mL
A10644-01
Collagen I, Coated Plate 6-well
5 plates
A11428-01
Collagen I, Coated Plate 24-well
5 plates
A11428-02
Collagen I, Coated Plate 96-well
5 plates
A11428-03
Instruments
Countess® Automated Cell Counter (includes 50 Countess® cell
counting chamber slides and 2 mL of Trypan Blue Stain)
1 unit C10227
Mallassez Hemocytometer 1 unit
99503
Neon™ Transfection System 1 unit
MPK5000
Qubit® Fluorometer 1 unit
Q32857
*Some of the products listed in the table are also available in different quantities and packaging sizes.
Growth Factors and
Purified Proteins
Life Technologies offers an array of highly-potent and highly-pure growth factors,
chemokines, cytokines, and other proteins and protein inhibitors validated for use in cell
culture. These products have been validated in live cell bioassays using GIBCO
®
media.
For more information and a complete list, refer to www.invitrogen.com/cellculture.

Cell Culture Basics | 51
Part 5. Appendix
Transfection and
Selection
Researchers have a wide variety of gene delivery techniques to introduce plasmid DNA,
siRNA or duplex RNAi, oligonucleotides, and RNA into eukaryotic cells for a variety of
research and drug discovery applications. For more information on the most appropriate
transfection method for your cell line and application, refer to our Transfection Selection
Guide.
Plasmid Transfection
Life Technologies offers the most complete collection of cationic lipid-based transfection
reagents with exceptional performance that can be used for plasmid delivery into a broad
range of cells. Lipofectamine
™
LTX with PLUS
™
Reagent (Cat. no. 15338-100) is a plasmid
DNA-specific transfection reagent that provides maximum expression with minimum
cytotoxicity.
RNAi Transfection
RNA interference (RNAi) technology is revolutionizing the biological discovery process
as well as target discovery and validation. Using RNAi, you can turn gene expression
“off”, or knock it down, to better understand its function and role in disease. High-
efficiency transfection is an essential first step for achieving effective gene knockdown.
Life Technologies offers the most complete collection of cationic lipid-based transfection
reagents with exceptional performance that can be used for delivery of assorted RNAi
reagents, including shRNA and miR RNAi vectors and synthetic molecules such as
siRNA, Stealth RNAi
™
siRNA, and Dicer-generated siRNAi pools. Further, cell specific
RNAi transfection protocols have been developed using these transfection reagents for
many popular cell lines. For more information and a complete list, refer to
www.invitrogen.com/transfection.
Transient Protein Production and High Throughput Screening
For the highest protein yields in mammalian suspension cells, FreeStyle
™
MAX
Transfection Reagent (Cat. no. 16447-100) is a versatile and scalable transient expression
tool for CHO and 293 suspension cell cultures. FreeStyle
™
MAX Reagent is a proprietary,
animal origin-free formulation for the transfection of plasmid DNA into eukaryotic cells
that can easily be scaled up to produce large amounts of recombinant proteins. FreeStyle
™
MAX Reagent allows the highest expression levels and transfection rates with lowest
cytotoxicity in bio-production applications.
Viral Delivery Systems
Having evolved to proficiently deliver nucleic acids to cells, viruses offer a means to
reach hard-to-transfect cell types for protein overexpression or knockdown. Adenoviral,
oncoretroviral, and lentiviral vectors have been used extensively for delivery in cell
culture and in vivo. Life Technologies offers a variety of products used in viral-mediated
transduction, including BLOCK-iT
™
Adenoviral and Lentiviral Expression Systems. For
more information, refer to www.invitrogen.com.
Neon
™
Transfection System
The Neon
™
Transfection System (Cat. no. MPK5000) is a novel, benchtop electroporation
device that employs an electroporation technology by using the pipette tip as an
electroporation chamber to efficiently transfect mammalian cells including primary and
immortalized hematopoietic cells, stem cells, and primary cells. The Neon
™
Transfection
System efficiently delivers nucleic acids, proteins, and siRNA into all mammalian cell
types including primary and stem cells with a high cell survival rate.

52 | Cell Culture Basics
Part 5. Appendix
Transfection
Reagents
For most applications, including plasmid DNA and siRNA transfections, Lipofectamine
™
2000 Transfection Reagent provides the best efficiency and gene expression with
maximum viability across a broad range of adherent and suspension cell lines. In addition
to Lipofectamine
™
2000, Life Technologies has additional reagents which may better suit
your experiments. Consult the information below for your particular application.
Transfection Reagent Cat. no. Key Features and Applications of Reagent
Lipofectamine™ LTX 15338-500 • Provides high transfection eciency and signicantly lower
toxicity levels for a wide range of cell lines
• Provides signicantly improved transfection performance in a
number of primary and hard-to-transfect cell lines
• Optimized protocols for over 30 dierent cell lines are
available so much less time is needed for evaluation and
optimization
Lipofectamine™ LTX
and PLUS Reagent
15338-100
FreeStyle™ MAX 16447-100
16447-500
16447-750
• Optimized for transient transfection in CHO suspension cells
and also works for HEK-293 cells
• Used for a large-scale transient protein production with
milligrams of protein yield
Lipofectamine™
RNAiMAX
13778-075
13778-150
• Superior transfection eciency requiring lower RNAi
concentrations leading to more eective gene knockdown
with minimal non-specic eects
• Easy optimization due to minimal cytotoxicity across a
10-fold concentration range of transfection reagent
• Compatibility with a broad range of cell types providing
the most versatile approach to all of your gene silencing
experiments
Lipofectamine™ 2000 11668-019
11668-027
11668-500
• Achieves highest eciency and expression results for
plasmid or RNAi transfections
• Works eectively with many cell types (adherent or
suspension)
• Easy and fast protocol – no need for wash steps before or
after transfection
• Complexes can be added to cells growing in serum-
containing media
• Ideal if transfecting at conuencies of 90% or greater
(minimizes cytotoxicity following transfection)
Lipofectamine™ 2000
CD
12566-014 Same performance as Lipofectamine™ 2000, certied animal-
origin free (“CD” = chemically dened)
Oligofectamine™ 12252-011 Transfection of antisense oligonucleotides
293fectin™ 12347-019
12347-500
12347-750
• Used for transient protein production in combination with
the FreeStyle™ 293 Expression System
• Optimized for suspension FreeStyle™ 293-F cells
Cellfectin® II 10362-100 Optimal transfection of insect cells, including S2, Sf9, Sf21 and
High Five™ cells
DMRIE-C 10459-014 Transfection of suspension cells, including CHO, lymphoid and
Jurkat cell lines
Optifect™ 12579-017 • Broad use reagent designed for low conuency applications
(<70% conuent at the time of transfection)
• Useful for cell lines that are sensitive to transfection reagents

Cell Culture Basics | 53
Part 5. Appendix
Additional Resources
Mammalian and
Insect Cell Cultures
For more information on mammalian and insect cell culture, cell type specific protocols,
and additional cell culture products, refer to Mammalian Cell Culture and Insect Cell
Culture portals on our website.
Cell and Tissue
Analysis
Understanding the structural and functional relationships of cells and tissues is critical
to advancements in key research disciplines, including molecular biology, genetics,
reproductive function, immunology, cancer and neurobiology. Key components of cell
and tissue analysis are cell viability and proliferation, cell signaling pathways, cell cycle
analysis, and cell structure. Life Technologies has a broad portfolio of reagents and kits
for cell and tissue analysis, including Molecular Probes
®
Fluorescence Products and
technologies as well as Dynal
®
Bead-based Solutions for cell isolation and expansion.
From antibodies and stem cell research products to benchtop instruments like the
Countess
™
Cell Counter and Qubit
™
Fluorometer, Life Technologies has the tools essential
for cellular analysis research. For more information, refer to the Cell & Tissue Analysis
portal on our website.
Transfection
Selection Tool
Transfection of DNA, siRNA, oligonucleotides, and RNA into mammalian cells is a critical
step in many research efforts of the biomedical scientific community including gene
silencing using RNAi, transient gene expression studies, protein/antibody production,
and generation of stable cell lines. To simplify the transfection optimization process,
Invitrogen, part of Life Technologies, has created a transfection selection tool that provides
information on recommended transfection reagents with optimized protocols for over 120
cell lines. Invitrogen offers a wide range of transfection reagents for your specific needs.
For more information and cell type specific protocols, refer to Transfection Selection Tool
on our website.
Safety Data Sheets Safety Data Sheets (SDS) are available at www.invitrogen.com/sds.
Certificate of
Analysis
The Certificate of Analysis provides detailed quality control and product qualification
information for each product. Certificates of Analysis are available on our website. Go to
www.invitrogen.com/support and search for the Certificate of Analysis by product lot
number, which is printed on the box.
54 | Cell Culture Basics
Part 5. Appendix
Technical Support
World Wide Web Visit the Invitrogen website at www.invitrogen.com for:
• Technical resources, including manuals, vector maps and sequences, application notes,
SDSs, FAQs, formulations, citations, handbooks, etc.
• Complete technical support contact information
• Access to the Invitrogen Online Catalog
• Additional product information and special offers
Contact Us For more information or technical assistance, call, write, fax, or e-mail. Additional
international offices are listed on our website (www.invitrogen.com).
Corporate Headquarters Japanese Headquarters European Headquarters
5791 Van Allen Way
Carlsbad, CA 92008 USA
Tel: 1 760 603 7200
Tel (Toll Free): 1 800 955 6288
Fax: 1 760 602 6500
E-mail:
LOOP-X Bldg. 6F
3-9-15, Kaigan
Minato-ku, Tokyo 108-0022
Tel: 81 3 5730 6509
Fax: 81 3 5730 6519
E-mail:
jpinfo@invitrogen.com
Inchinnan Business Park
3 Fountain Drive
Paisley PA4 9RF, UK
Tel: +44 (0) 141 814 6100
Tech Fax: +44 (0) 141 814 6117
E-mail:
eurotech@invitrogen.com
Limited Warranty Invitrogen (a part of Life Technologies Corporation) is committed to providing our
customers with high-quality goods and services. Our goal is to ensure that every
customer is 100% satisfied with our products and our service. If you should have any
questions or concerns about an Invitrogen product or service, contact our Technical
Support Representatives. All Invitrogen products are warranted to perform according
to specifications stated on the certificate of analysis. The Company will replace, free
of charge, any product that does not meet those specifications. This warranty limits
the Company’s liability to only the price of the product. No warranty is granted for
products beyond their listed expiration date. No warranty is applicable unless all product
components are stored in accordance with instructions. The Company reserves the
right to select the method(s) used to analyze a product unless the Company agrees to a
specified method in writing prior to acceptance of the order. Invitrogen makes every effort
to ensure the accuracy of its publications, but realizes that the occasional typographical
or other error is inevitable. Therefore the Company makes no warranty of any kind
regarding the contents of any publications or documentation. If you discover an error in
any of our publications, please report it to our Technical Support Representatives.
Life Technologies Corporation shall have no responsibility or liability for any special,
incidental, indirect or consequential loss or damage whatsoever. The above limited
warranty is sole and exclusive. No other warranty is made, whether expressed or
implied, including any warranty of merchantability or fitness for a particular purpose.

Cell Culture Basics | 55
Part 5. Appendix
Notes

www.invitrogen.com/cellculturebasics
B-087243 0110
CELL CULTURE BASICS
Handbook
Cell Culture Basics
Cell Culture Basics
