
April 2023
Public Health Data Goals and
2-Year Milestones
Public Health
Data Strategy

2
Executive
summary
1. Ensure Core Data Sources are more complete, timely, rapidly exchanged, and available to support the integrated ability to detect, monitor, investigate, and
respond to public health threats
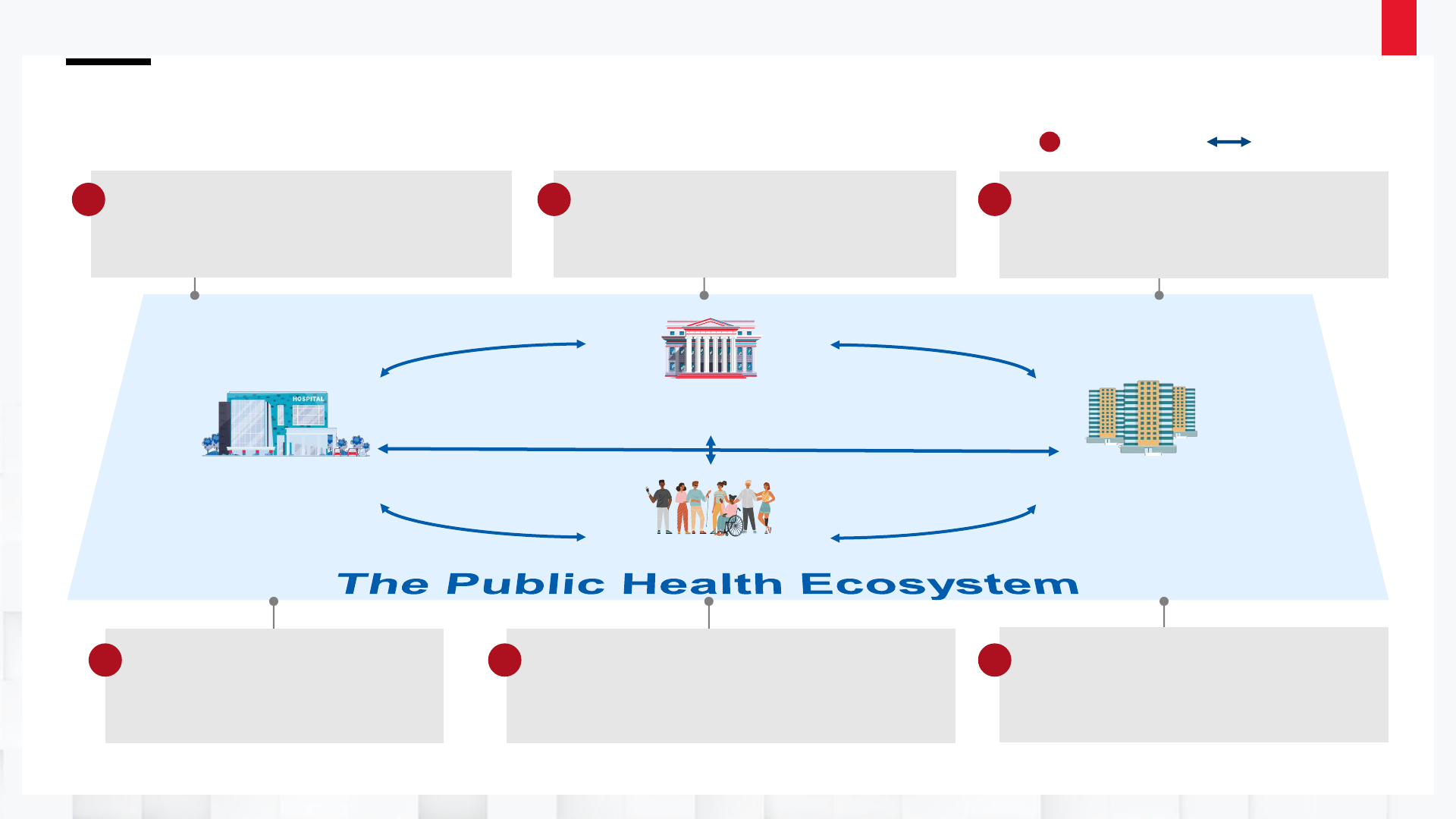
The Public Health Data
Strategy (PHDS) outlines the
data, technology, policy,
and administrative actions
essential to exchange
critical core data efficiently
and securely across
healthcare and public health.
The strategy is designed to
describe a path to address
gaps in public health data,
helping the nation become
response-ready, promote
health equity, and improve
health outcomes for all.
To advance core public health missions (Detect and Monitor, Investigate and Respond, Inform
and Disseminate, and Be Response-Ready), the PHDS addresses the CDC Moving Forward
imperative to consistently deliver public health information and guidance to Americans in near
real-time.
Through the PHDS, public health decision-makers (e.g., the public; labs; providers; other
healthcare partners; state, tribal, local, and territorial health departments; CDC programs; federal
agencies) will have a clear, streamlined vision of near-term priorities.
The Public Health Data Strategy outlines four Public Health Data Goals:
1. Strengthen the core of public health data
1
2. Accelerate access to analytic and automated solutions to support public health investigations
and advance health equity
3. Visualize and share insights to inform public health action
4. Advance more open and interoperable public health data
Accomplishing 2-year milestones associated with these goals requires collaboration and
partnership with state, tribal, local, and territorial health departments; healthcare partners; and
other federal agencies.
CDC will create accountability for these Public Health Data Goals and 2-year milestones
through the new Office of Public Health Data, Surveillance, and Technology (OPHDST),
which will lead and strengthen engagement and collaboration with jurisdictions, CDC programs,
and private partners, and focus the execution of data modernization activities.

4
What the Public Health Data Strategy is
The Public Health Data Strategy (PHDS) outlines the data,
technology, policy, and administrative actions essential to
exchange critical core data efficiently and securely
across healthcare and public health.
The strategy is designed to describe a path to address gaps in
public health data, helping the nation become response-ready,
promote health equity, and improve health outcomes for all.

5
Why the Public Health Data Strategy matters
To advance core missions of robust public
health data aimed at improving health
outcomes equitably…
Detect and
monitor
Investigate
and respond
Inform and
disseminate
Be response-
ready
…the Public Health Data Strategy …
Addresses the imperative of the CDC Moving Forward effort
to consistently deliver public health information and guidance to
Americans in near real-time
Measures success with specific 2-year milestones
Aligns data modernization efforts at all levels of public health
and across partners, focusing on near-term priorities
Creates accountability for public health data with CDC's
newly established Office of Public Health Data, Surveillance, and
Technology
Builds on lessons learned from the COVID-19 pandemic and
other recent public health threats to be more response-ready
Source: CDC Moving Forward website
6
The Public Health Data Strategy aims to address challenges currently experienced
across healthcare and public health
ILLUSTRATIVE
Healthcare
(labs, providers)
National public health
(CDC, Federal partners)
Public
States, tribes, localities, and territories
(STLTs)
12+ months for data on some reportable conditions
to become available in national datasets or be
disseminated in accessible and interoperable
formats
4
~3 months between first reported domestic
mpox case and CDC data access
agreements with STLTs
5
6+ months often needed to develop
and potentially rework Data Use
Agreements
4
30%+ of COVID-19 cases missing data on
race and ethnicity early in the pandemic
3
~70% of healthcare organizations using fax to
send or receive care records
1
Up to 80% of epidemiologists’ time spent
cleaning data
2
because of non-interoperable
systems
! ! !
! ! !
Example challenge
!
High-level data flow
1. ONC Data Brief No. 54 (2021), 2. ‘A Prototype of Modernized Public Health Infrastructure for All: Findings from a Virginia Pilot’ – CDC (2022), 3. CDC case surveillance data (as of Sept 9, 2022), 4. Average estimates by CDC staff, 5. ‘Very
Harmful’ lack of data blunts U.S. response to outbreaks’ – New York Times 2022
Source: New York Times, NEJM Jan 2022, GAO, ONC, CDC + USDS Virginia prototype findings, CDC estimates

7
Successfully achieving the Public Health Data Goals and 2-year milestones
addresses critical public health challenges by 2025
Public health data goal supported
#
Healthcare
(labs, providers)
National public health
(CDC, Federal partners)
Public
States, tribes, localities, and territories
(STLTs)
Increased ability for STLTs to automatically
exchange data with CDC creates a reliable
national common operating picture
Data on demand enables health
disparities analysis across geographies,
conditions, and settings
Faster sharing of data through
language and terms for data
protection and use
<7 days needed to detect a suspected
disease outbreak and begin nationwide
monitoring
2
American public has near real-time awareness of
the status of high-consequence diseases
through a centralized data dissemination
platform
3
1
4 1
Time saved on data cleaning and
analytics, through reusable technologies,
enables epidemiologists to focus on core
public health missions
2
ILLUSTRATIVE
High-level data flow

8
The Public Health Data Strategy supports partners across the public health ecosystem
1. STLT: States, Tribes, Localities, and Territories
Federal agencies
Understand where and how to access richer public health
data on demand to inform decision-making
…To…
The Public Health Data
Strategy will help…
The Public
Have greater access to critical information on public health
emergencies, risks, trends, and resources
STLTs
1
Prioritize data and technology investments to enable the
most critical public health systems to be scalable, flexible,
interoperable, sustainable, reusable, and intuitive
Streamline ongoing and planned efforts to support
measurable and concrete 2-year milestones
CDC programs
Healthcare
(labs and providers)
Identify and adopt ready-to-use tools that enable easier
and faster sharing of critical core public health data
Enabling
greater
response
readiness
and
progress
toward
health
equity

9
Public Health Data Strategy strengthens the core of public health data
Core Data Sources
1
are essential to identify diseases and conditions, detect emerging public health threats, and understand
disease burden and severity across different populations. The PHDS decreases the burden to securely report these critical data,
and improves dissemination to ensure the right data are available at the right time.
Case
Emergency
dept.
Immun-
ization
Vital stats.
Healthcare
capacity
Lab
Core Data Sources…
Case data represent comprehensive disease and condition information used by public health to understand
disease burden, know who is at risk, and identify outbreaks
Laboratory data, including test results and test type, enable public health agencies to track disease trends and
identify outbreaks or exposures, and help frontline providers diagnose and treat health conditions
Emergency department data, including clinical diagnoses, signs, and symptoms, help identify near real-time
trends for new, emerging, and developing public health threats to inform faster detection and response
Vital statistics data include birth and death data and are essential to understand disease severity, mortality,
trauma, and toxicity that might signal a larger public health emergency
Immunization data capture vaccine doses administered (both routinely recommended and response-related)
to support calculating vaccination coverage levels and trends
Healthcare capacity and utilization data assess availability of healthcare resources, including staff, beds, and
equipment, aiding understanding of health system stresses and disease severity to inform resource allocation
1. Core Data Sources as defined in CDC Advisory Committee to the Director (ACD) Data and Surveillance Workgroup (DSW) Report; non-exhaustive of all data sources critical to public health
awareness and response (e.g., advanced molecular detection data)

11
Four major Public Health Data Goals enable the core public health missions
Detect and
monitor
Investigate
and respond
Inform and
disseminate
Be response-
ready
Make tools available so STLTs and other public health decision-makers can better use
public health data to address health disparities
1
2
Public Health Data Goals
Core public
health missions
1. Case (including electronic case reporting [eCR]), lab (including electronic lab reporting [ELR], Electronic Test Orders and Results [ETOR]), emergency department (including National Syndromic Surveillance Program [NSSP] emergency
department data), vital statistics, immunization, healthcare capacity (including National Healthcare Safety Network [NHSN] data)
Ensure Core Data Sources
1
are more complete, timely, rapidly exchanged, and available to
support the integrated ability to detect, monitor, investigate, and respond to public health threats
3
Serve as a trusted source for near real-time visualizations and offer situational awareness for
the public and decision-makers to understand risks, make decisions, and direct resources
4
Enable exchange of interoperable data so that healthcare, STLTs, federal agency partners,
and CDC programs can access and use data they need, when they need it
Strengthen the core of public health data
Accelerate access to analytic and automated solutions to support public health
investigations and advance health equity
Visualize and share insights to inform public health action
Advance more open and interoperable public health data

12
Public Health Data Goals drive key outcomes over the next 2 years
Goals
Accelerate access to analytic
and automated solutions to
support public health
investigations and advance
health equity
2
STLTs and CDC can identify emerging threats in all parts of the nation in a timely way as more critical access hospitals in rural communities
send case data electronically
STLTs can address gaps in the public health workflow such as linking case, lab, and immunization records to enrich data and inform public
health action through reusable technologies
STLTs and communities can identify, prevent, and mitigate disproportionate impact on populations through use of automated reports
provided by CDC, using CDC public health databases
Visualize and share
insights to inform public
health action
3
STLTs and CDC programs receive quicker access to data and insights from Core Data Sources
1
Americans can access near real-time data and visualizations made available via a centralized data dissemination platform (e.g., a
Public Health Data Channel like the National Weather Service) to improve understanding and inform decisions about public health risks
Advance more open and
interoperable public
health data
4
CDC establishes a strategic pathway of data exchange with providers’ electronic health records (EHRs) through at least 2 initial
Trusted Exchange Framework and Common Agreement (TEFCA) for public health use case pilots
CDC and STLTs increase data exchange back to healthcare providers, helping to inform clinical decision-making
Data use and access are easier through established, standardized agreements (e.g., for sharing emergency department data with CDC
programs, STLTs), enabling quicker access to minimal data necessary for response during a public health emergency
CDC has measurably and securely increased the number of accessible open public health data sets for timely use and faster insights
Strengthen the core of
public health data
1
States, tribes, localities, and territories (STLTs) have reduced reporting burden as case data are electronically reported in a consistent
format, using a CDC Front Door concept
Public health labs can forecast needs better with integrated, electronic ability to see test orders before samples arrive and ensure faster
exchange of orders and test results with submitters
STLTs can access lab reports more quickly and through multiple pathways, enabling faster public health action (e.g., case
investigation, contact tracing)
CDC can access lab, case, and mortality data faster, enabling robust situational awareness that informs decision-making across the
nation
STLTs and CDC programs have access to more early warning signals from emergency departments (EDs), with up to 80% participation
by US non-federal EDs in the National Syndromic Surveillance Program (NSSP)
1. Case (including electronic case reporting [eCR]), lab (including electronic lab reporting [ELR}, Electronic Test Orders and Results [ETOR]), emergency department (including National Syndromic Surveillance Program [NSSP] emergency department visit data), vital statistics,
immunization, healthcare capacity (including National Healthcare Safety Network [NHSN] data)
By the end of 2024, 2-year milestones strive to ensure…
Accomplishing the Public Health Data Goals requires collaboration and partnership with STLTs, healthcare partners, and other federal agencies
SPECIFIC MILESTONES BY YEAR ON FOLLOWING PAGES
ILLUSTRATIVE

13
Success is measured by 2-year milestones (for Goal 1)
Public Health Data
Goal
Milestones within 2 years
1
Strengthen the
core of public
health data
Ensure Core Data
Sources
2
are more
complete, timely,
rapidly exchanged,
and available to
support the
integrated ability to
detect, monitor,
investigate, and
respond to public
health threats
1
End of 2024 End of 2023
38 jurisdictions³ are ingesting eCRdata into disease surveillance systems32 jurisdictions³ are ingesting eCR data into disease surveillance
systems
Almost 100%of ELC recipients are connected to multiple intermediaries (e.g.,
AIMS, ReportStream, HIEs) for lab data
90% of ELC recipients are connected to one or multiple intermediaries
(e.g., AIMS, ReportStream, HIEs) for lab data
100% of State Public Health Labs have implemented ETOR (e.g., web portal,
direct integration, or use of intermediary) with at least 1 healthcare partner for at
least 1 lab program
90% of State Public Health Labs have implemented ETOR (e.g., web
portal, direct integration, or use of intermediary) with at least 1
healthcare partner for at least 1 lab program
Core case data for select nationally notifiable conditions are reported using a
common format, using a CDC Front Doorconcept, and shared back in near
real-time for CDC programs and STLT partners to access
STLTs enabled to submit a generic core case data feed that can be
used for national disease notification
50% of lab test order requests received electronically at CDC infectious
disease labs(e.g.,using ETOR, CSTOR, intermediary)
75% of CDC infectious disease labs send lab test results to external
partners electronically (e.g., using ELR, CSTOR, intermediary)
Increased participation to 80% (from 73% today) of U.S. non-federal
emergency departments to increase representativeness of NSSP data sources
and users
Reduced time to send mortality data to and receive coded cause of
death data from CDC for 30 additional jurisdictions (42–45 total)³
through use of FHIR messaging
Reduced time to send mortality data to and receive coded cause of
death data from CDC for 12–15 jurisdictions³ through use of FHIR
messaging
1. Accomplishing the Public Health Data Goals requires collaboration and partnership with STLTs, healthcare partners, and other federal agencies 2. Case (including electronic case reporting [eCR]), lab (including electronic lab reporting [ELR], Electronic Test Orders and Results [ETOR]), emergency department (including
National Syndromic Surveillance Program [NSSP] emergency department data), vital statistics, immunization, healthcare capacity (including National Healthcare Safety Network [NHSN] data) 3. Out of the ~64 ELC- or PHI- funded jurisdictions
Potential impact: <7 days needed to detect a suspected disease outbreak and begin nation-wide monitoring, through using faster case, lab, emergency department, mortality data
CDC receives and ensures access to commercial lab data from at least
3 major national and regional commercial labs to enable situational
awareness across multiple conditions
CDC receives and ensures access to commercial lab data from at least
2 major national commercial labs to enable situational awareness
across multiple conditions

14
Success is measured by 2-year milestones (for Goal 2)
At least 2 automated reports using CDC’s
healthcare databases available within CDC
and across STLTs to identify and address
health disparities
Public Health Data
Goal
Accelerate access
to analytic and
automated
solutions to
support public
health
investigations and
advance health
equity
Make tools available
so STLTs and other
public health
decision-makers
can better use
public health data to
address health
disparities
2
End of 2024 End of 2023
Critical access hospitals in production with
eCR increased to 25%, up from 20% in 2022
Critical access hospitals in production with
eCR increased to 35%
Reusable technologies to link multiple data
streams (e.g., case, lab) made available to all
jurisdictions and deployed by at least 1 STLT
– saving time for epidemiologists who currently
spend up to 80% of their time on data cleaning
2
Reusable technologies to link multiple data
streams (e.g., case, lab) adopted by multiple
STLTs
Milestones within 2 years
1
1. Accomplishing the Public Health Data Goals requires collaboration and partnership with STLTs, healthcare partners, and other federal agencies
2. ‘A Prototype of Modernized Public health Infrastructure for all: Findings from a Virginia Pilot’ – CDC (2022)
Potential impact: Improved data available to epidemiologists (e.g., 2x critical access hospitals sending case data electronically), enabling faster health equity analyses
15
Success is measured by 2-year milestones (for Goal 3)
Minimum viable product for centralized
data dissemination platform developed in
partnership with CDC’s Office of Readiness
and Response to share timely and actionable
data publicly
Centralized data dissemination platform
launched to streamline insights from case,
emergency department, mortality data, and at
least 1 other data source
Data and visualizations available within 2–3
days (from 5–90+ days) for CDC programs
and STLTs since time of receiving case data
at CDC for at least 1 nationally notifiable
condition (i.e., viral hepatitis)
Public Health Data
Goal
Visualize and
share insights to
inform public
health action
Serve as a trusted
source for near real-
time visualizations
and offer situational
awareness for the
public and decision-
makers to
understand risks,
make decisions,
and direct
resources
3
End of 2024 End of 2023
Data and visualizations available within 2–3
days (from 5–90+ days) for CDC programs
and STLTs since time of receiving case data
at CDC for multiple nationally notifiable
conditions
Milestones within 2 years
1
1. Accomplishing the Public Health Data Goals requires collaboration and partnership with STLTs, healthcare partners, and other federal agencies
Potential impact: Faster, actionable insights available to the public in near real-time (2–3 days from 5–90+ days) through a centralized data dissemination platform

16
Success is measured by 2-year milestones (for Goal 4)
Public Health Data
Goal
Advance more
open and
interoperable
public health data
Enable exchange of
interoperable data
so that healthcare,
STLTs, federal
agency partners,
and CDC programs
can access and use
data they need,
when they need it
4
End of 2024 End of 2023
New data access agreement established to enable
easier sharing of emergency department data from
NSSP across STLTs and CDC programs
At least 50% of existing NSSP jurisdictions adopt new
data access agreement to enable easier sharing of
emergency department data across STLTs and CDC
programs
Number of public health data sets published by
CDC with metadata utilizing FAIR open data
principles increased by 10%
Milestones within 2 years
1
1. Accomplishing the Public Health Data Goals requires collaboration and partnership with STLTs, healthcare partners, and other federal agencies
Minimal data elements necessary for public health
response defined for at least case and lab data, in
collaboration with STLT partners and CDC programs
Minimal data elements necessary for public health
response defined for multiple data sources, in
collaboration with STLT partners and CDC programs
CDC launches pilots for at least 2 public health
use cases with TEFCA (e.g., query data from
healthcare settings for urgent public health
investigations)
CDC selects a QHIN and has identified at least 2
public health use cases for TEFCA, establishing
a pathway for data exchange with healthcare
systems and providers
Standard language and terms for data
protection and use agreed upon with public health
partners for Core Data Sources, consistent with
ACD DSW recommendations
Data access and use under established language
and terms across at least 15% of funded states
and territories for Core Data Sources, including
case data
Potential impact: Standardized language and terms for data use introduced across healthcare and public health, enabling greater data quality and easier data sharing

18
How CDC will help achieve the Public Health Data Strategy goals and
milestones
Increase engagement and collaboration with:
STLTs: Collaborate on and continually provide feedback on progress towards key 2-year milestones (e.g., DUAs,
reusable technologies); engage directly with STLTs to understand priority needs
Providers and labs: Gather feedback from the frontlines on how CDC can better support progress towards 2-year
milestones
CDC: Establish internal steering committee to provide guidance on longer term PHDS, monitor and provide input on
progress; establish mechanisms to solicit programmatic input and identify priority needs
Private partners: Organize Industry Days and promote data exchange pilots to work toward modernization together
Federal agency partners: Collaborate with partners such as ONC and CMS to advance shared understanding of
activities needed to support 2-year milestones, including TEFCA for public health
Increase engagement, collaboration, and continual feedback
Ensure accountability for the Public Health Data Goals sits with CDC’s Office of Public Health Data, Surveillance, and
Technology (OPHDST):
Structure the organization to support core public health missions
Identify owners within OPHDST and across the agency to drive progress for specific 2-year milestones
Update the agency regularly on Public Health Data Strategy goals and milestones
Establish accountable office within CDC

19
The Public Health Data Strategy is an ambitious but necessary plan
to improve the exchange of core data across healthcare and public
health.
Successful implementation of the strategy—and achievement of the
Public Health Data Goals over the next two years―will require
collaboration with STLTs, healthcare partners, and other federal
agencies, as well as sustained resources.

20
List of terms used in this document
State Public Health LaboratorySPHL
States, Tribes, Localities, and Territories STLT
TEFCA Trusted Exchange Framework and Common Agreement
Association of Public Health Laboratories (APHL) Informatics Messaging Services platform
AIMS
CDC Specimen Test Order and ReportingCSTOR
Data Use AgreementDUA
Electronic Case ReportingeCR
Electronic Health RecordsEHR
Epidemiology and Lab Capacity Cooperative AgreementELC
Electronic Laboratory ReportingELR
Electronic Test Orders and ResultsETOR
Findability, Accessibility, Interoperability, and Reuse (of digital assets)FAIR
Health Information Exchange HIE
NEDSS Base SystemNBS
National Healthcare Safety NetworkNHSN
National Syndromic Surveillance Program NSSP
Public Health Data StrategyPHDS
Qualified Health Information NetworkQHIN
Fast Healthcare Interoperability ResourcesFHIR
Office of the Chief Information OfficerOCIO
Data Modernization InitiativeDMI
Objectives and Key ResultsOKR
Advisory Committee to the Director (ACD), Centers for Disease Control and Prevention
ACD
Data and Surveillance Workgroup (within the Advisory Committee to the Director)DSW



